jorden7544
10.07.2019 •
Chemistry
Asupposedly silver crown is tested to determine its density. it displaces 10.7 ml of water and has a mass of 112 g. part a could the crown be made of silver? could the crown be made of silver? no, the crown did not make of silver. the obtained density is higher than the density of silver which is 10.5 g/ml. yes, the crown made of silver. the obtained density is equal to the density of silver which is 10.5 g/ml. no, the crown did not make of silver. the obtained density is lower than the density of silver which is 10.5 g/ml.
Solved
Show answers
More tips
- S Style and Beauty How to Properly Tie a Tie: 5 Simple Steps...
- L Leisure and Entertainment How to Make a Crab Trap in Just a Few Minutes...
- H Health and Medicine How Much Does Abortion Cost? Expert Answers and Insights...
- S Sport How to Build Arm Muscles? Effective Exercises and Tips...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
Answers on questions: Chemistry
- C Chemistry Suspension mixture or puse substance 1. copper 2. sodium chloride 3. muddy water 4. hot coffee 5. mill. cassification of matter 29os copper mixture or a pure substance ?...
- M Mathematics megan feeds her dog 1/2 cup of food each morning and each evening. how much food does she feed him in a 7 day week...
- M Mathematics Please help I need it like nowwww...
- M Mathematics (x - 1)/(n - 1) + (2n ^ 2 * (1 - x))/(n ^ 4 - 1) = (2x - 1)/(1 - n ^ 4) - (1 - x)/(1 + n) alguien me puede ayudar con este problema es una ecuacion de 1er grado...

Ответ:
Density of a substance is the mass per unit volume of that substance. Density values of a substance are constant.
Given the mass of the crown = 112 g
Volume of water displaced by the crown = 10.7 mL
Density of the crown =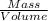
=
The crown is supposed to be made of silver. The density of silver is 10.5g/mL. The calculated value of density from given mass and volume displaced by the crown is equal to the theoretical density value of silver.
So the correct answer will be that the crown is made of silver as the estimated density is equal to that to density of silver.
Ответ:
you can put this one: The children ate the picnic enthusiastically while the birds pecked gladly at the breadcrumbs.
Explanation: