dtaylor7755
25.03.2020 •
Chemistry
At equilibrium, a 4.50 L container has 2.6 g of carbon, CO2 at a partial pressure of 0.0020 atm, and a total pressure of 0.572 atm. Calculate KP for this reaction at 725oC.
Solved
Show answers
More tips
- S Science and Technology How to Secure Exam Sessions: Silence Mobile Phones in the Classroom...
- G Goods and services How to Choose a Bread Maker?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- E English Hunting is a rural sport population growth in rural areas increases the pressure against hunting. what is the correct way? a. hunting is a rural sport population...
- M Mathematics 6 plus y as a algebraic expression...
- M Mathematics Solve the following system of equations by linear combination: 2d + e = 8 d – e = 4 select one: a. the solution is (5, –2). b. the solution is (4, 0). c. there is...
- M Mathematics A rectangle has an area of x^2+6x+8. What are the dimensions of the rectangle?...

Ответ:
The given question is incomplete.. The complete question is :
Consider the following reaction:
At equilibrium, a 4.50 L container has 2.6 g of carbon, CO2 at a partial pressure of 0.0020 atm, and a total pressure of 0.572 atm. Calculate KP for this reaction at 725oC.
162.45
Explanation:
Equilibrium pressure of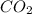 = 0.0020 atm
= 0.0020 atm
Total pressure at equilibrium = 0.572 atm
Equilibrium pressure of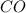 = Total pressure at equilibrium - Equilibrium pressure of
= Total pressure at equilibrium - Equilibrium pressure of 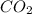 = (0.572- 0.0020) = 0.570 atm
= (0.572- 0.0020) = 0.570 atm
The given balanced equilibrium reaction is,
At eqm 0 0.0020 0.570
The expression for equilibrium constant for this reaction will be,
Now put all the given values in this expression, we get :
By solving we get :
Thus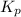 for this reaction at
for this reaction at 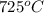 is 162.45
is 162.45
Ответ:
CALCULATION:
For the reaction
Ca(OH)2 → Ca2+ + 2OH-
we will use the Ksp expression to solve for the concentration [OH-] and then use the acid base concepts to get the pH:
Ksp = [Ca2+][OH-]^2
The listed Ksp value is 5.5 x 10^-6. Substituting this to the Ksp expression, we have
Ksp = 5.5 x 10^-6 = (s) (2s)^2 = 4s^3
s3 = 5.5x10^-6 / 4
Taking the cube root, we now have
s = cube root of (5.5x10^-6 / 4)s
= 0.01112
We know that the value of [OH-] is actually equal to 2s:
[OH-] = 2s = 2 * 0.01112 = 0.02224 M
We can now calculate for pOH:
pOH = - log [OH-]
= -log(0.02224)
= 1.65
Therefore, the pH is
pH = 14 - pOH
= 14 - 1.65
= 12.35