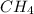B.) what fraction of a mole is 10g of clindamycin?
Solved
Show answers
More tips
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- D Dating, Love, Relationships How Long Can Love Last?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- F Food and Cooking How to Find Your Zip Code?...
- S Style and Beauty How to choose the best mascara for your eyelashes...
- S Style and Beauty How to Apply Foundation Correctly?...
- S Sport How to Choose Tennis Rackets?...
Answers on questions: Chemistry
- C Chemistry Stewardship is the application of various methods that use up natural resource.. true or false ?...
- C Chemistry Assuming your average walking speed is 1.4 m/s, what time should you leave your house to arrive at school (0.84 km away) at 7:15?...
- C Chemistry State a living thing in which each type of fermentation could take place...
- C Chemistry As the concentration of a given salt solution increases, the conductivity of the solution: a) increases b) no way to predict c) remains the same d) increases then decreases decreases...
- C Chemistry What is hydrolysis,and what type of weathering is it...
- C Chemistry How many electrons would a neutral atom of nitrogen need to lose in order to have a full valence electron shell? a. 3 b. 5 c. 7 d. more information is needed to figure this out....
- C Chemistry Explain how oxygen is connected to chemical weathering...
- C Chemistry Is liquid starch with water a suspension, colloid, or solution?...
- C Chemistry Calculate the average atomic mass of unknown element X. You determine that isotope 127X makes up 80%, isotope 126X makes up 17%, and isotope 128X makes up 3%. C. a. 127.10 amu b. 126.86...
- C Chemistry (same table as the last question) Three students measured the length of a stamp that has is exactly 2.71 cm long and recorded their measurements in the table below. What is the relative...

Ответ:
10 g / 424.98 (grams per mole) = .024 moles of clindamycin
Ответ:
445.15 kJ of heat could be obtained by the combustion of 8.0 grams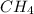
Explanation:
To calculate the moles :
The given thermochemical equation is:
According to stoichiometry :
1 mole of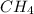 on combustion produce heat = 890.3 kJ
on combustion produce heat = 890.3 kJ
Thus 0.50 moles of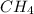 on combustion produce heat =
on combustion produce heat =
Thus 445.15 kJ of heat could be obtained by the combustion of 8.0 grams