LiannaMarquez542
03.09.2019 •
Chemistry
Balance the following ionic equation for a redox reaction, using whole number coefficients. mno−4(aq)+so2−3(aq)+h3o+(aq)⟶mn2+(aq)+so2−4(aq)+h2o(l)
Solved
Show answers
More tips
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...

Ответ:
Answer : The balance ionic equation for a redox reaction will be,
Explanation :
The half balanced redox reactions in acidic medium will be:
(1)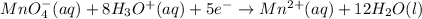
(2)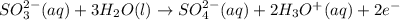
In order of balance the electrons, we multiply the equation 1 by 2 and equation 2 by 5, we get:
(1)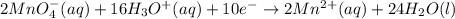
(2)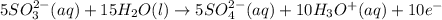
Now adding both the equation, we get:
The overall reaction will be,
Ответ:
yup
Explanation: