ashcormu11
18.10.2020 •
Chemistry
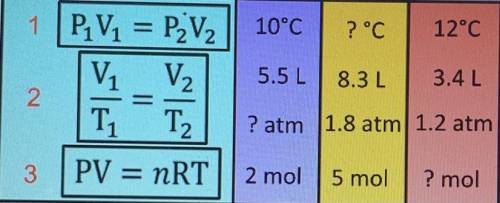
Based on the three formulas shown, use one of them to solve for the purple yellow and red box and explain how you did it.
Solved
Show answers
More tips
- F Food and Cooking What age is appropriate for giving your child cocoa?...
- A Auto and Moto How to Start a Diesel Engine in Cold Weather?...
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- S Sport How to Do a Jumping Split...
- H Health and Medicine How Did Inna Lose Weight on Dom 2?...
- F Family and Home How to Properly Fold Napkins in a Napkin Holder?...
- F Food and Cooking How to Set Up Ventrilo - The Ultimate Guide...
- S Science and Technology How to Make a Homemade Smoker: The Ultimate Guide...
- A Auto and Moto Battle for the Relocation of The Cherkizovsky Market: Who Won?...
Answers on questions: Chemistry
- C Chemistry Which of the following processes has directly caused ocean currents to change over earth s history? a. ice-albedo feedback b. continental drift c. axial precession d. volcanic...
- C Chemistry What is the layer of the earths atmosphere that we live in...
- C Chemistry . i have some question on alef website...
- C Chemistry HELP HELP HELP how are scientist using computer models to simulate worldwide ocean activity...
- C Chemistry Which statement supports the following conclusion? Sheep were the best choice for the first domesticated livestock. Sheep descended from a wild species in Europe and Asia....
- C Chemistry An atom that has 13 protons and 15 neutrons is element 10.81 +3 12.011 4 14.0067 3 15.9994 2 18.9984 20.180 B C CN O F Ne 8 9 5 2-3 26.98154 16 2-4 131 28.0855 10 2-8 139.948...
- C Chemistry Which of the following statements is incorrect: A. A combustion reaction is a reaction where diatomicoxygen is a product B. When the combustion ofa reactant that contains...
- C Chemistry Acetic acid and ethanol react to form ethyl acetate and water like this: HCH3CO2(aq) + C2H5OH(aq) - C2H5CO2CH3(aq) + H2O(l). Imagine 244 mol of HCH3CO2 are added to a flask...
- C Chemistry How much work does the climber do on his pack if his pack weighs 90 N and he climbs to a height of 30 m?...
- M Mathematics Join zoom ID: 990 3441 4563 PASS: At4t6u...

Ответ:
P = 11.133 atm (purple)
T = -236.733 °C(yellow)
n = 0.174 mol(red)
Further explanationSome of the laws regarding gas, can apply to ideal gas (volume expansion does not occur when the gas is heated),:
Boyle's law at constant T, P = 1 / V Charles's law, at constant P, V = T Avogadro's law, at constant P and T, V = nSo that the three laws can be combined into a single gas equation, the ideal gas equation
In general, the gas equation can be written
where
P = pressure, atm
V = volume, liter
n = number of moles
R = gas constant = 0.08206 L.atm / mol K
T = temperature, Kelvin
To choose the formula used, we refer to the data provided
Because the data provided are temperature, pressure, volume and moles, than we use the formula PV = nRT
Purple boxT= 10 +273.15 = 373.15 K
V=5.5 L
n=2 mol

Yellow boxV=8.3 L
P=1.8 atm
n=5 mol

Red boxT = 12 + 273.15 = 285.15 K
V=3.4 L
P=1.2 atm
Ответ: