PONBallfordM89
07.09.2019 •
Chemistry
Before the introduction of chlorofluorocarbons, sulfur dioxide (enthalpy of vaporization, 5.96 kcal/mol) was used in household refrigerators. what mass (in kg) of so2 must be evaporated to remove as much heat as evaporation of 1.24 kg of ccl2f2 (enthalpy of vaporization is 20.1 kj/mol)?
Solved
Show answers
More tips
- H Health and Medicine Which Water Are You Drinking? Is it Worth Buying Bottled Water?...
- A Animals and plants Уход за джунгариками: полезные советы и рекомендации...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
Answers on questions: Chemistry
- H History An example of a service industry job in an urban center?...
- M Mathematics Repersent 3 and 1/3 on a number line...
- H History Which of the following events contributed to the start of the modern era...
- M Mathematics A student carries a backpack that weighs 10 pounds. They also carry an instrument that weighs x pounds. Which equation represents the total weight?...
- H History What is the authority to hear a case before any other case a. jurisdiction b. original jurisdiction c. judicial review d. judicial authority...

Ответ:
0.5286 kilogram of sulfur dioxide must be evaporated .
Explanation:
Enthalpy of vaporization of chlorofluorocarbons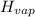 = 20.1 kJ/mol
= 20.1 kJ/mol
Mass of chlorofluorocarbons = 1.24 kg = 1240 g
Moles of chlorofluorocarbons =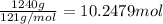
Heat removes by 10.2479 moles of chlorofluorocarbons : q
Let moles of sulfur dioxide removing 205.983 kJ(q) of heat be n.
Enthalpy of vaporization of sulfur dioxide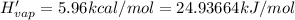
(1 kcal = 4.184 kJ)
n = 8.2603 moles
Mass of 8.2603 moles of sulfur dioxide =
0.5286 kilogram of sulfur dioxide must be evaporated .
Ответ:
If you're asking what I think you're asking, the answer is yes.
Explanation
It may take people who drink alcohol often 4-5 drink to be drunk, versus somebody who almost never drinks 1 drink to be drunk. People start to build a tolerance the more they drink/take something. Same with spicy food and drugs. This can become a problem, as addicts may start to drink more and more to get drunk. This can cause serious damage to the body.