blakestuhan
05.09.2019 •
Chemistry
Brass is a substitutional alloy consisting of a solution of copper and zinc. a particular sample of red brass consisting of 79.0% cu and 21.0% zn by mass has a density of 8740 kg/m3.what is the molality and molarity of zn in the solid solution?
Solved
Show answers
More tips
- O Other Everything You Need to Know About Kudyabliks...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- F Food and Cooking Homemade kvass: recipe and brewing process...
- H Health and Medicine How to Choose the Right Tanning Cream?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- S Sport Running: How to Do It Right?...
- H Health and Medicine Sunstroke: Causes, Symptoms, and Precautions...
- H Health and Medicine How to Deal with Heat Stroke?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- A Auto and Moto How many blood alcohol level units are allowed in Russian traffic laws?...
Answers on questions: Chemistry
- C Chemistry When burning wood, some of the mass of the wood seems to disappear. Where does that mass go? It is destroyed. O It is recombined into gases like carbon dioxide....
- C Chemistry Which one of the following is not a double-displacement reaction?...
- C Chemistry Which of these is true of chemical reactions? A.) all of these are true B.) In a chemical reaction, one or more substances undergo a change in state. C.) In a chemical reaction,...
- C Chemistry En la cristalizacion aumenta o disminuye el calor...
- C Chemistry JOIN MY MEETING NOWID-9384026088PASSWORD 1234...
- C Chemistry Grams of oxygen to create 36 4 grams of hydrogen reacts with grams of water. 40 O 32 09 O 8...
- C Chemistry Is anyone good at chemistry if so can someone help me please ? If I have 5 moles of a gas at a pressure of 6.5 atm and a volume of 14 liters, what is the temperature? PV=nRT...
- C Chemistry A fatty acid that has a many hydgrogen atoms is called?...
- C Chemistry What is the ph of a 0.033 m koh solution...
- C Chemistry Consider the sublimation of iodine at 25.0 °c : i2(s) ¡ i2( g) a. find δg° rxn at 25.0 °c. b. find δgrxn at 25.0 °c under the following nonstandard conditions: i. pi2 =...

Ответ:
The molality and molarity of zinc in the solution is 4.07 m and 28.11 M respectively.
Explanation:
Solute is the substance which is present in smaller proportion in a mixture and solvent is the substance which is present in larger proportion in a mixture.
We are given:
(m/m) % of Cu = 79 %
This means that 79 g of copper is present in 100 grams of alloy.
(m/m) % of Zn = 21 %
This means that 21 g of zinc is present in 100 grams of alloy.
As, zinc is present in smaller proportion. So, it is solute and copper is the solvent.
Calculating molality of zinc:To calculate molality of the zinc, we use the equation:
where,
Putting values in above equation, we get:

Calculating molarity of zinc:To calculate volume of a substance, we use the equation:
Density of solution =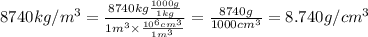
(Conversion factors used are: 1 kg = 1000 g & )
)
Mass of solution = 100 g
Putting values in above equation, we get:
To calculate the molarity of solution, we use the equation:
We are given:
Molar mass of zinc = 65.3 g/mol
Volume of solution = (Conversion factor:
(Conversion factor:  )
)
Mass of zinc = 21.0 g
Putting values in above equation, we get:
Hence, the molality and molarity of zinc in the solution is 4.07 m and 28.11 M respectively.
Ответ:
1. Plasma is "the state..." because plasma is rarely natural on earth (only lightning strikes) and has the highest temp
gas is "particles move freely" they do move freely
liquid is "it expands to fill volume" they do not have definite boundaries so they fill up the container
solid is "it retains its shape" yup dis is pretty obvious ig
2.
I agree with your answers.
it wouldnt be hypothesis or theory because they are basically the same thing and they explain facts. fact would be the best answer
conduct experiments would be the most logical here because you can gain new knowledge from conducting experiments instead of the rest.