Morghan7153
31.08.2019 •
Chemistry
By titration it is found that 68.5 ml of 0.154 m naoh(aq) is needed to neutralize 25.0 ml of hcl(aq). calculate the concentration of the hcl solution.
Solved
Show answers
More tips
- C Computers and Internet Отправляем смс через интернет: легко и просто...
- H Health and Medicine Want to Lose Weight? Here s How Many Calories You Need Per Day...
- H Health and Medicine How Many Ribs Do Humans Have?...
- C Computers and Internet Dropbox: What is it and How to Use it...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- A Auto and Moto How many blood alcohol level units are allowed in Russian traffic laws?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- H Health and Medicine How to Deal with Heat Stroke?...
- H Health and Medicine Sunstroke: Causes, Symptoms, and Precautions...
Answers on questions: Chemistry
- C Chemistry Explain the energy transformations involved when the plate subducts and creates an earthquake...
- C Chemistry Where does the koala belong in the cladogram shown below? g placental limbs jaws segmented body multicellular unicellular common ancestor...
- C Chemistry Draw the major organic product formed from the reaction below. when an organic species is an ion, do not show the counter ion (i.e., do not show the spectator ion.) (a) draw the...
- C Chemistry Compounds that contain a fused ring system are called these have three 6-membered rings and one 5-membered ring. some of these compounds are found in biological membranes. are the...
- C Chemistry Which statement best describes the process of the formation of the solar system?...
- C Chemistry Critical thinking : Place an unmagnetized piece of iron in a magnetic field (eg iron filings near a magnet). Why is it attracted to the magnet?...
- C Chemistry Piense en los metales que estudió en el laboratorio práctico. ¿Qué partículas subatómicas crees que influyeron más en la reactividad? ¿Cómo podría el número de partículas subatómicas...
- C Chemistry If a overscript right-arrow endscripts minus b overscript right-arrow endscripts = 3.8c overscript right-arrow endscripts, a overscript right-arrow endscripts plus b overscript...
- M Mathematics Arecipe calls for 3 3/4 cups of flour how much flour is needed to make 1/2 the recipe...
- B Business The legal documents that identify the basic agreements between partners are called:...

Ответ:
The concentration of 25 mL of HCl solution needed to neutralize 68.5 mL of 0.154 M NaOH is 0.422 M
We'll begin by calculating the number of mole of NaOH in the solution. This can be obtained as follow:
Volume of NaOH = 68.5 mL = 68.5 / 1000 = 0.0685 L
Molarity of NaOH = 0.154 M
Mole of NaOH =?Mole = Molarity × Volume
Mole of NaOH = 0.154 × 0.0685
Mole of NaOH = 0.010549 moleNext, we shall determine the number of mole of HCl needed to react with 0.010549 mole of NaOH. This can be obtained as follow:
NaOH + HCl ⇒ NaCl + H₂O
From the balanced equation above,
1 mole of NaOH reacted with 1 mole of HCl.
Therefore,
0.010549 mole of NaOH will also react with 0.010549 mole of HCl.
Finally, we shall determine concentration of the HCl solution. This can be obtained as follow:
Volume of HCl = 25 mL = 25 / 1000 = 0.025 L
Mole of HCl = 0.010549 mole of HCl.
Molarity of HCl =?Molarity = mole / Volume
Molarity of HCl = 0.010549 / 0.025
Molarity of HCl = 0.422 MTherefore, the concentration of the HCl solution needed for the reaction is 0.422 M
Learn more: link
Ответ:
The concentration of the HCl solution : 0.421 m
Further explanationTitration is a procedure for determining the concentration of a solution by reacting with another solution which is known to be concentrated (usually a standard solution). Determination of the endpoint/equivalence point of the reaction can use indicators according to the appropriate pH range
Titrations can be distinguished including acid-base titration, depositional titration, and redox titration. An acid-base titration is the principle of neutralization of acids and bases is used.
An acid-base titration there will be a change in the pH of the solution.
From this pH change a Titration Curve can be made which is a function of acid / base volume and pH of the solution
Acid-base titration formula
Ma. Va. na = Mb. Vb. nbMa, Mb = acid base concentration
Va, Vb = acid base volume
na, nb = acid base valence
Neutralization Reaction :
NaOH + HCl ⇒ NaCl + H₂O
68.5 ml of 0.154 m NaOH
25.0 ml of HCl
Acid-base titration formula
Ma. Va. na = Mb. Vb. nb
a = NaOH, b = HCl(both have valence 1)
0.154 m . 68.5.1 = Mb . 25 . 1
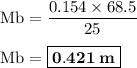
Learn morethe best tool to find the volume of that she needs to neutralize the acid
link
Determine the endpoint of the titration.
link
acid-base
link
Identify the acids and the bases
link
endpoint titration
link
Ответ:
A) A very thin atmosphere and temperatures wary from 427 degrees to -189 degrees Celsius (800 to -292 degrees Fahrenheit).
Explanation: