sharperenae463
02.03.2020 •
Chemistry
Calculate Kc for the reaction: 2 HI(g) ⇄ H2(g) + I2(g) given that the concentrations of each species at equilibrium are as follows: [HI] = 0.85 mol/L, [I2] = 0.60 mol/L, [H2] = 0.27 mol/L.
Solved
Show answers
More tips
- C Computers and Internet Are there special gaming mice?...
- L Leisure and Entertainment When will Maslenitsa start?...
- F Food and Cooking Discovering the Mysterious Fruit of Feijoa...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
Answers on questions: Chemistry
- B Biology What are three limited resources...
- A Arts If yes, can you share with us how was your experience...
- H History Why did many colonist protest the stamp act?...
- M Mathematics A survey of magazine subscribers placed 59 celebrities in 3 categories. 35 were on the most powerful list. 26 were on the most liked list. 20 were on the most intelligent...
- M Mathematics What is 12/7 please help and i will be very happy...

Ответ:
The value of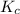 for the given reaction is 0.224
for the given reaction is 0.224
Explanation:
For the given chemical equation:
The expression of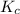 for given equation follows:
for given equation follows:
We are given:
Putting values in above expression, we get:
Hence, the value of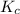 for the given reaction is 0.224
for the given reaction is 0.224
Ответ: