naiomireyes74p2aybs
18.06.2021 •
Chemistry
Calculate the amount of heat needed to melt 160. g of solid octane (C8H18 ) and bring it to a temperature of 99.2 degrees c. Round your answer to 3 significant digits. Also, be sure your answer contains a unit symbol.
Solved
Show answers
More tips
- C Computers and Internet Can You Insert a Regular SIM Card into an iPad? Let s Find Out...
- F Food and Cooking Лечо: вкусное и простое блюдо для любой кухни...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
Answers on questions: Chemistry
- C Chemistry How many moles are present in a sample if it consists of 5.61x10^24 particles?...
- C Chemistry Hey help me please I need to get this grade...
- C Chemistry Hydogen can be produced according to the following word equation. Complete a balance chemical equation for this chemical reaction :Zinc + Hydrochloric =Zinc chloride + hydrogen...
- C Chemistry The graph represents velocity vs. time for carla...
- C Chemistry Fill in the blank with the term that best completes each sentence. During the life cycle of a seedless plant, a sporophyte releases . When spores land, they grow into gametophytes....
- C Chemistry Which characteristic is a behavioral adaptation of the Venus fly trap? roots that the plant uses to obtain water leaves that close when touched flowers to attract pollinators leaves...
- C Chemistry Different individuals within a population have developed two ways of escaping a predator. One has become fast and sleek and uses great amounts of energy escaping the predator. The...
- C Chemistry 9. Use the equation below to answer the question. HCI + NaOH NaCl + H20 If 50 g of HCl is combined with 40 g of NaOH, which is the limiting reactant? O NaOH Онсі...
- C Chemistry how could a change to the size of the walleye pollock population causes an increase in the size of the moon population Please answer I m confused...
- C Chemistry Match the following activities. Click the item in the left column. Use the plus sign to move it up or the minus sign to move it down until it matches the correct entry in the right...

Ответ:
The amount of heat needed to melt the given amount of octane is 84.6 kJ
Explanation:
We know:
Boiling point of Octane =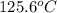
Few processes involved are:
(1):
(2):
Calculating the heat absorbed for the process having same temperature:
where,
q is the amount of heat absorbed, n is the moles of sample and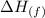 is the enthalpy of fusion
is the enthalpy of fusion
Calculating the heat released for the process having different temperature:
where,
n = moles of sample
The number of moles is defined as the ratio of the mass of a substance to its molar mass.
The equation used is:
Given mass of octane = 160. g
Molar mass of octane = 114.23 g/mol
Plugging values in equation 3:

For process 1:We are given:
Putting values in equation (i), we get:

For process 2:We are given:
Putting values in equation (ii), we get:
Calculating the total amount of heat released:
Hence, the amount of heat needed to melt the given amount of octane is 84.6 kJ
Ответ:
True
Explanation:
This is true because every chemical reacts differently