deannabrown2293
29.11.2020 •
Chemistry
Calculate the change in entropy when 10.0 g of CO2 isothermally expands from a volume of 6.15 L to 11.5 L. Assume that the gas behaves ideally.
Solved
Show answers
More tips
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- O Other What is a Disk Emulsifier and How Does it Work?...
- S Sport How to Pump Your Chest Muscle? Secrets of Training...
- C Computers and Internet How to Get Rid of 3pic Infector: Everything You Need to Know...
- S Style and Beauty How to Grow Hair Faster: Real Methods and Advice...
- C Computers and Internet How to Top Up Your Skype Account Without Losing Money?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
Answers on questions: Chemistry
- H History Drag each tile to the correct box. Match the civil rights leader with the organization. Helen Keller W. E. B. Du Bois Henry Gerber NAACP ACLU Society for Human Rights Reset Nec...
- P Physics Is a caterpillar turning into a butterfly is or is not a metamorphosis...
- M Mathematics A student charges $31.50 for 3 hours of babysitting. If the amount the student earns babysitting is proportional to the hours spent babysitting, which equation models the amount...
- H History 1) which of these changes during the industrial revolution led to greater efficiency? a) workers received labor rights. b) major labor unions were formed. c) slavery and other...

Ответ:
The change in entropy of the carbon dioxide is kilojoules per Kelvin.
kilojoules per Kelvin.
Explanation:
By assuming that carbon dioxide behaves ideally, the change in entropy ( ), measured in kilojoules per Kelvin, is defined by the following expression:
), measured in kilojoules per Kelvin, is defined by the following expression:
Where:
If we know that ,
, 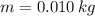 ,
,  ,
,  ,
, 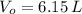 and
and  , then the change in entropy of the carbon dioxide is:
, then the change in entropy of the carbon dioxide is:
The change in entropy of the carbon dioxide is kilojoules per Kelvin.
kilojoules per Kelvin.
Ответ:
cation, 2+
a cookie made from flour, eggs, sugar, butter, and chocolate chips.
melting point is a physical property of matter because when a substance melts, it changes state from a solid to a liquid but is still the same substance.
melting point is the temperature at which a substance changes from a solid to a liquid state. it is a physical property because measuring melting point does not change the identity of the substance.
a potassium ion will form a 1+ charge because it will have more protons than electrons.
the electrons would repel each other because they have the same charge.
explanation: