kristadwsn
23.08.2019 •
Chemistry
Calculate the electric double layer thickness of a alumina colloid in a dilute (0.1 mol/dm3) csci electrolyte solution at 30 °c. how are these numbers affected by the addition of 0.1 mol/dm3 of kcl? at what distance from the particle surface (r) has the potential decayed to 1% of its initial value?
Solved
Show answers
More tips
- H Health and Medicine How Did Inna Lose Weight on Dom 2?...
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: Chemistry
- E Engineering Describe the different types of at-grade intersections. Also give an example of an appropriate location for the use of each type....
- M Mathematics Which portion of the unit circle satisfies the trigonometric inequality cos? + sin? 22 1? Assume that e is the angle made by the positive x-axis and a ray from the...
- M Mathematics The expression 4x* represents 144...
- B Business The top ten arms-exporting companies give millions to...

Ответ:
Explanation:
The given data is as follows.
Concentration = 0.1
= 0.1 \frac{mol dm^{3}}{dm^{3}} \frac{10^{3}}{dm^{3}} \times \frac{6.022 \times 10^{23}}{1 mol} ions
=
T = = (30 + 273) K = 303 K
= (30 + 273) K = 303 K
Formula for electric double layer thickness ( ) is as follows.
) is as follows.
where, = concentration =
= concentration = 
Hence, putting the given values into the above equation as follows.
=
= m
m
or, =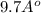
= 1 nm (approx)
Also, it is known that =
= 
Hence, we can conclude that addition of 0.1 of KCl in 0.1
of KCl in 0.1  of NaBr "
of NaBr " " will decrease but not significantly.
" will decrease but not significantly.
Ответ:
Answer :
In my opinion , answer is A