GreenHerbz206
20.06.2020 •
Chemistry
Calculate the EMF between copper and silver Ag+e-E=0.89v
Cu=E=0.34v
Solved
Show answers
More tips
- F Family and Home Stay Warm but Don t Overheat: What is the Optimal Temperature for Your Home During Winter?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
Answers on questions: Chemistry
- M Mathematics F(11)=7(x-1)+8 what’s the value of 11...
- M Mathematics To test the effectiveness of a new drug, a researcher gives one group of patients the new drug and another group a placebo. Results are shown below:MedicationEffectiveNot effectiveActive...
- P Physics Aball is thrown vertically upwards with an initial velocity of 20.00 m/s. neglecting air resistance, how long is the ball in the air? what is the greatest height reached by the ball?...
- M Mathematics CLASSWORK: Factoring Quadratics where a 1 2.) Factoring Quadratics 2x² + 6x - 8 First Middle Last Term Term Term Steps 3.) Factoring Quadratics 3x? + 7x - 30...

Ответ:
Depending on the value of
value of 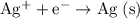 , the cell potential would be:
, the cell potential would be:
Explanation:
In this galvanic cell, the following two reactions are going on:
The conversion betweenNote that the standard reduction potential of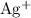 ions to
ions to  is higher than that of
is higher than that of 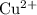 ions to
ions to  . Alternatively, consider the fact that in the metal activity series, copper is more reactive than silver. Either way, the reaction is this cell will be spontaneous (and will generate a positive EMF) only if
. Alternatively, consider the fact that in the metal activity series, copper is more reactive than silver. Either way, the reaction is this cell will be spontaneous (and will generate a positive EMF) only if 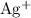 ions are reduced while
ions are reduced while  is oxidized.
is oxidized.
Therefore:
The reduction reaction at the cathode will be:The cell potential is the sum of the electrode potentials at the cathode and at the anode:
Using data from the 1985 and 2012 CRC Handbook:
Ответ:
Formic acid is the stronger acid.
Explanation:The strength of an acid is determined by the power of it to dissociate and produce H⁺ ions. Greater the dissociation of an acid, the stronger that acid is. Also, extent of dissociation for particular acid is given by its dissociation constant i.e. Ka.
The greater the Ka value, the stronger an acid is and vice versa.
Calculating Ka value of Pivalic acid:
As,
pKa = -log [Ka]
So,
Ka = 10^-pKa
Putting value of pKa,
Ka = 10⁻⁵
Ka = 1.0 × 10⁻⁵
Calculating Ka value of Formic acid:
As,
pKa = -log [Ka]
So,
Ka = 10^-pKa
Putting value of pKa,
Ka = 10⁻³'⁸
Ka = 1.58 × 10⁻⁴
Conclusion:As the Ka value of Formic acid is greater than Pivalic acid therefore, it is stronger acid than Pivalic acid.
TIP:When comparing strength of acids, the acid with the lowest pKa value is considered the strongest acid and vice versa.