Calculate the mass of magnesium carbonate ( MgCO3), in grams, required to produce 110.0 g of carbon dioxide using the following equation: MgCO3 --> MgO CO2
Solved
Show answers
More tips
- C Computers and Internet How Much Does an iPhone Cost in America?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- D Dating, Love, Relationships How Long Can Love Last?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- F Food and Cooking How to Find Your Zip Code?...
- S Style and Beauty How to choose the best mascara for your eyelashes...
- S Style and Beauty How to Apply Foundation Correctly?...
Answers on questions: Chemistry
- C Chemistry 7. What causes pressure in a closed container of gas?...
- C Chemistry Fill in the chart listed down below...
- C Chemistry What is the mass of chlorine in 3.9 x 1019 molecules of Cl2?...
- C Chemistry The heat of vaporization of a liquid is 1300 J/g. What is the minimum number of joules needed to change 70.0 g of the liquid to vapor at the boiling point?...
- C Chemistry Be sure to answer all parts. Take all the thermodynamic data from your textbook. Assume the data do not vary with temperature. (a) Calculate ΔSuniv for the reaction H2O2(l)...
- C Chemistry Describe the motion of molecules in an ice cube and in a radiator in winter. Molecules in an ice cube move more slowly than molecules radiator COMPLETE Describe the motion...
- C Chemistry What animals are in the Arctic Tundra?...
- E English 9. Which of the following is true for sentences that have more than one clause? OA There can be two negative words per clause. OB There should never be more than one negative...
- M Mathematics find the area of a rectangle whose length is 8 and breath is 3...
- S Social Studies 1. Most of the people like to live in a. mountains b. plains c. plateaus 3. The areas with harsh and unbearable climate conditions are populated a. densely b. moderately...

Ответ:
Explanation:
We have to start with the reaction:
We have the same amount of atoms on both sides, so, we can continue. The next step is to find the number of moles that we have in the 110.0 g of carbon dioxide, to this, we have to know the atomic mass of each atom:
C: 12 g/mol
O: 16 g/mol
Mg: 23.3 g/mol
If we take into account the number of atoms in the formula, we can calculate the molar mass of carbon dioxide:
In other words: . With this in mind, we can calculate the moles:
. With this in mind, we can calculate the moles:
Now, the molar ratio between carbon dioxide and magnesium carbonate is 1:1, so:
With the molar mass of (
(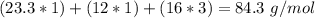 . With this in mind, we can calculate the grams of magnesium carbonate:
. With this in mind, we can calculate the grams of magnesium carbonate:
I hope it helps!
Ответ:
hm
Explanation: