joannegrace4898
10.07.2019 •
Chemistry
Calculate the mass of magnesium sulfate heptahydrate required to prepare 0.100 dm3 of a 0.200 mol dm^-3 solution
Solved
Show answers
More tips
- S Science and Technology How to Check a Barcode: Unique Product Identifier...
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry Question 2 1 pts Calculate the mass of solute, in grams, required to prepare 28.0 mL of 0.170 M cupric sulfate...
- C Chemistry Harrison is making a cake. He prepares some batter and places it into the oven. Harrison realize there’s a gas, carbon dioxide, coming out of the batter as it bakes. Which...
- C Chemistry Why an offspring and its parent share many common traits?...
- C Chemistry Caco3(s) ∆→cao(s) + co2(g). if 13.2 g of co2 was produced from the thermal decomposition of 40.1 g of caco3, what is the percentage yield of the reaction?...
- E English I m sure every diver dreams about the Titanic, said the speaker at the Sea Rovers meeting. To dive down to that fabulousship would be the ultimate thrill in underwater exploration....
- B Business A health and fitness center wants to grow and expand its services but at the same time sees the best way to reduce costs is by trimming the number of trainers and staff it...
- B Business When goods that have already been approved for purchase are received but have not yet been paid for, which of the following local government accounts should be increased?...
- C Chemistry When KCl dissolves in water A) the Clions are attracted to dissolved K+ ions. B) the Clions are attracted to the partially negative oxygen atoms of the water molecule. C)...
- E English Find the figures of speech in this poem. Don t let people bring you down, everyone is perfect in their own way and never forget that because people that try to bring you...
- M Mathematics Which equation demonstrates the multiplicative identity property? ( -3 + 5 i) + 0 = -3 + 5 i ( -3 + 5 i ) (1) = -3 + 5 i ( -3 + 5 i) ( -3 + 5 i) = - 16 - 30 i ( -3 + 5 i)...

Ответ:
- 4.93 grams of magnesium sulfate heptahydrate.
Solution:- We have been given with molarity and volume of the solution and asked to calculate the grams of the solute required to make the given solution.
From given volume and molarity we could calculate the moles of the solute and on multiplying with the molar mass of the compound we get the grams of it.
Magnesium sulfate heptahydrate formula is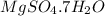 ,
,
Molar mass of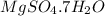 = 24.31 + 32.06 + 4(16) + 14(1.0) + 7(16)
= 24.31 + 32.06 + 4(16) + 14(1.0) + 7(16)
= 24.31 + 32.06 + 64 + 14 + 112
= 246.37 gram per mol
Let's make the set up using dimensional set up as:
= 4.93 grams
Hence, 4.93 grams of magnesium sulfate heptahydrate are required to make of
of  solution.
solution.
Ответ: