Calculate the mass of oxygen needed to produce 10.0 grams
of CO2.
Please help
Solved
Show answers
More tips
- C Computers and Internet Dynamically Assigned IP Address: What Is It and How Does It Work?...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
Answers on questions: Chemistry
- C Chemistry Which groups of elements are not included in the alien periodic table? do you think it is likely that an alien planet would lack these elements? explain....
- H History What is the role of artists in freedom fighting (example: musicians, painters, writers, etc...)...
- M Mathematics The length of michelle s room is shown. the ratio of length to width is 4: 3. what is the width of michelle s room...
- M Mathematics (02.02 MC) What set of reflections would carry triangle ABC onto itself? Which is the correct label of the parallel lines?(4 points)x-axis, y=x, y-axis, x-axisx-axis, y-axis,...

Ответ:
The mass of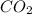 and
and  produced are 336.6 g and 183.6 g respectively.
produced are 336.6 g and 183.6 g respectively.
Explanation:
The combustion reaction between propane and oxygen leads to formation of carbon dioxide and water.
Law of Conservation of mass states that the mass will remain constant for a balanced equation. This is carried out when the total number of atoms on reactant side is same as the total number of atoms on the product side. Thus the equation must be balanced.
a) 1 mol of propane produces = 3 moles of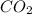
Thus 2.55 mol of propane produces =![\frac{3}{1}\times 2.55=7.65 moles of [tex]CO_2](/tpl/images/1130/4175/dfc34.png)
mass of
b) 1 mol of propane produces = 4 moles of
Thus 2.55 mol of propane produces =![\frac{4}{1}\times 2.55=10.2 moles of [tex]H_2O](/tpl/images/1130/4175/74c43.png)
mass of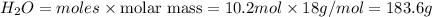
The mass of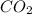 and
and  produced are 336.6 g and 183.6 g respectively.
produced are 336.6 g and 183.6 g respectively.