BakedBiscuit6896
17.07.2020 •
Chemistry
Calculate the mass of sodium hydroxide in 30cm3 of a 0.105 mol/dm3 solution
Solved
Show answers
More tips
- C Computers and Internet Best Applications for Your iPad: Review of the Best Candidates for Installation...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
Answers on questions: Chemistry
- C Chemistry LiOH + KCl → LiCl + KOH The reaction above should theoretically yield 35.5 grams of LiCl. When Tom actually completes the reaction in the lab, he produced only 6 grams...
- C Chemistry If you have a pressure of 350 Torr and volume of 200 mliters what would the new volume be if the pressure was changed to 700 Torr...
- C Chemistry The results of an analysis are 66.97g, compared to the accepted value 67.06g. Calculate the absolute error and the relative errors in parts per thousand...
- C Chemistry JOIN MY MEETINGID-9384026088PASSWORD 1234...
- C Chemistry 3. Write the half reaction that describes the reduction reaction that occurs when iron corrodes in air and water...
- C Chemistry How do i balance equations like this?...
- C Chemistry Chemical energy. chemica Fuels contain a store of energy called When fuels are burned the energy. Some fuels, called were formed from the remains of plants or energy...
- C Chemistry If 0.50 g of copper metal was reacted with excess nitric acid, determine the mass of copper (II) nitrate, nitrogen dioxide gas, and water. Cu (s) + 4HNO3 (aq) → Cu(NO3)2...
- C Chemistry Which elements belong to the same group? N, P, As,Sb Ne, F, Ar,Kr Rb, Sr, Y, Zr K, Ca, Mg, Be...
- C Chemistry Identify the type of reaction shown in the following equation C6H12O6 + 6O2 = 6CO2 + 6H2O...

Ответ:
44.04 g/mol
Explanation:
Ideal gas law in terms of density:
where,
P = pressure of the gas = 0.789 atm
V = volume of the gas
T = temperature of the gas =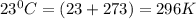
n = number of moles of the gas
R = gas constant = 0.0821 Latm/moleK
M = molar mass of gas
w = mass of gas
Thus molar mass of the gas is 44.04 g/mol