Calculate the molality of acetone in an aqueous solution with a mole fraction for acetone of 0.241. Answer in units of m.
Solved
Show answers
More tips
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- B Biology Acell is lacking in its supply of amino acids. which of the following will occur? a- mrna will not be formed because enzymes may not be present. b- the ribosomes will...
- B Business Bat Company s flexible budget for the units manufactured in May shows $15,710 of total factory overhead; this output level represents 70% of available capacity. During...
- B Business the u shape of the average total cost curve arises because of spreading total cost over a larger output and eventually returns....

Ответ:
The molality of solution is 17.6 mole/kg
Explanation:
Molality of a solution is defined as the number of moles of solute dissolved per kg of the solvent.
where,
n = moles of solute
moles of acetone (solute) = 0.241
moles of water (solvent )= (1-0.241) = 0.759
mass of water (solvent )=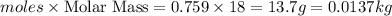
Now put all the given values in the formula of molality, we get
Therefore, the molality of solution is 17.6 mole/kg
Ответ:
give me a couple seconds and then we can chat
Explanation: