genyjoannerubiera
30.03.2020 •
Chemistry
Calculate the molarity of a solution containing 25.0 moles of ethanol (C₂H₅OH) in enough water to make 3.5 liters of solution?
Solved
Show answers
More tips
- S Style and Beauty What to Do When Hair Starts Falling Out?...
- H Health and Medicine Novomin: What is it and how to use it?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...

Ответ:
7.14mol/dm3
Explanation:
Molarity is calculated by dividing the amount of a substance in volume of the solution.
i.e Molarity = (Amount in moles/volume in liters or dm3)
Given that:
Amount of moles = 25.0 moles
Volume = 3.5 liters of solution
Recall that 1 liter = 1dm3
so, 3.5 liters = 3.5dm3
Molarity = (25.0moles/3.5dm3)
= 7.14mol/dm3
Thus, the molarity of the solution is 7.14mol/dm3
Ответ:
It will take 140.76 seconds to swim 0.150 miles.
Explanation:
Distance covered by man by swimming for t time= d
d = 1500.0 miles
t = 14 minutes + 34.56 seconds
1 min = 60 seconds
t = 14 × 60 seconds + 34.56 seconds = 874.56 seconds
Speed of the man while swimming = S =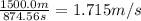
If the man swims 0.150 miles with the same speed ,then the time taken by him be T.
0.150 miles = 0.150 × 1609.34 m = 241.401 m
(1 mile= 1609.34 meters)
T = 140.76 seconds
It will take 140.76 seconds to swim 0.150 miles.