Deagon4434
13.04.2020 •
Chemistry
Calculate the specific heat capacity of titanium if a 43.56 g sample absorbs 476 J as its temperature changes from 20.13 oC to 41.06 oC. (0.522 J/g oC)
Solved
Show answers
More tips
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- S Sport How to Learn Swimming? Simple Tips for Beginners...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
Answers on questions: Chemistry
- C Chemistry In order for a liquid particle to become a gas it must: (choose all that apply) overcome the attractive forces of other liquid particles overcome the attractive forces...
- C Chemistry Kinetic energy is inversely proportional to mass and velocity directly proportional to mass and velocity directly proportional to mass and inversely proportional to velocity...
- C Chemistry Each component retains its own properties in a mixture. true or false...
- C Chemistry Please help with my homework about bedbugs!...
- C Chemistry Which item is NOT shown in a balanced chemical equation?...
- C Chemistry Question 4 Which of the following is a physical change? butane gas burns leaves are composted silver jewelry turns black o egg solidifies upon cooking fruit is dehydrated...
- C Chemistry Raoul ate a meal of oats before seeing his doctor. Oats are made of mostly starch molecules. Raoul feels tired, so his doctor gave him a test and found that Raoul’s cells...
- C Chemistry What is the % of water in (MgSO4 . 2H2O)?...
- C Chemistry A sample of an ideal gas has a volume of 2.29 L at 285 K and 1.12 atm. Calculate the pressure when the volume is 1.33 L and the temperature is 302 K....
- C Chemistry g Part A A Hofmann degradation of a primary amine forms an alkene that gives butanal and 2-methylpropanal upon ozonolysis and workup under reducing conditions. Identify...

Ответ:
The specific heat capacity of Titanium is found as 0.522 J / g°C.
Explanation:
We have to find the specific capacity using the formula as,
q = m × c × ΔT
Where,
q is the heat absorbed by the sample = 476 J
m is the mass of the sample = 43.56 g
c is the specific heat capacity of the sample = ?
ΔT is the temperature difference = 41.06° C - 20.13°C = 20.93° C
Now, we have to rewrite the equation to get the specific heat capacity as,
c =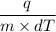
=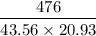
= 0.522 J / g°C
So the specific heat capacity of Titanium is found as 0.522 J / g°C.
Ответ:
Kerosene is lighter than water and so it floats on the water and continues to burn. Carbon (IV) oxide blankets the flame cutting off the supply of oxygen and extinguishing the fire.
Explanation:
As water is heavier than kerosene, it slips down permitting the kerosene to rise to the surface and continue to burn. Besides, the existing temperature is so high that the water poured on the fire would evaporate before it can extinguish the fire. Thus the kerosene fire cannot be extinguished with water.