hannahdrees0731
08.07.2021 •
Chemistry
Can someone put in the missing parts

Solved
Show answers
More tips
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- O Other What is a Disk Emulsifier and How Does it Work?...
- S Sport How to Pump Your Chest Muscle? Secrets of Training...
- C Computers and Internet How to Get Rid of 3pic Infector: Everything You Need to Know...
- S Style and Beauty How to Grow Hair Faster: Real Methods and Advice...
- C Computers and Internet How to Top Up Your Skype Account Without Losing Money?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
Answers on questions: Chemistry
- C Chemistry Students working in lab use 52.3g of CaCl2 to make a solution. When the solution is brought to a final volume of 800. mL, what is the concentration? A. 0.139 M B....
- C Chemistry Which pair of elements can be used to determine the age of a fossil that is over one billion years old?...
- C Chemistry How many mL of 8.00M HCl solution are needed to make a dilution with a final volume of 2,500.ml, and a concentration of HC-0.333M?...
- C Chemistry You decided to change your bedroom. You move your bed 1.5 meters across the room and the force you used to move the bed was 20 newtons. It takes you 5 minutes. How...
- C Chemistry The concentration of a solution is commonly expressed in terms of percent by mass or molarity. True or False?...
- C Chemistry I really need help please. NaOH + AlCl3 -- Al(OH)3 + NaCI. How many grams of AlCl3 are needed to produce 100.0 g NaCl?...
- C Chemistry conclu en expliquant pourquoi il n est pas intéressant d utiliser les éclairs comme sources d énergie....
- C Chemistry 3. Which of the following statements is not correct? A. All hybridization must involve an s-orbitalB. Excitation of Carbon atom in CH, involves promotion of one is...
- C Chemistry Engineers and designers must identify criteria and constraints and determine how these will affect the design process. Cite examples of how given criteria and constraints...
- C Chemistry A 2.584 g sample of a compound containing only carbon, hydrogen, and oxygen is burned in an excess of dioxygen, producing 5.874 g CO2 and 2.404 g H2O. What mass of...

Ответ:
1) 2.0 g
2) 0 g
3) 4.17 g
4) 2.57 g
Explanation:
First of all, we need to know the compounds and the reaction. The ion carbonate is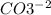 , and the ion nitrate is
, and the ion nitrate is 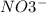 .
.
Sodium is in group 1, so it must lose one electron to be stable, and be the cation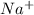 . Silver has only one electron too, so the cation will be
. Silver has only one electron too, so the cation will be 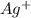 .
.
To form the chemical compounds, first we put the cation, then the anion, and change their charges without the signal:
Sodium carbonate: Na2CO3
Silver nitrate: AgNO3
Silver carbonate: Ag2CO3
Sodium nitrate: NaNO3
The balanced reaction will be:
Na2CO3 + 2 AgNO3 --> Ag2CO3 + 2 NaNO3
Now, we must check the stoichiometry, which will be 1:2:1:2 (always in number of moles)
The question wants to know the mass value, so we need to know the molar mass of these compounds. Checking the periodic table will see that:
Na = 23 g/mol, C = 12 g/mol, N = 14 g/mol, O = 16 g/mol, Ag = 108 g/mol
So the molar mass of the compounds must be:
Na2CO3 = 106 g/mol (2x23 + 12 + 3x16)
AgNO3 = 170 g/mol (108 + 14 + 3x16)
Ag2CO3 = 276 g/mol (2x108 + 12 + 3x16)
NaNO3 = 85 g/mol
We have a mixture of the reactants, so one probably would be in excess, so, first will need to test. Let's do the stoichiometry calculus using silver nitrate as the limit, so:
1 mol of Na2CO3 ---------- 2 mol of AgNO3
106 g ------------------------------ 2x170 = 340 g
x ------------------------------------ 5.14 g
By a simple direct three rule:
340x = 544.84
x = 1.6 g of Na2CO3
That means that for this reaction, we only need 1.6 g of Na2CO3 to react with 5.14 of AgNO3. How we have 3.60 g of Na2CO3, it is on excess, and all the AgNO3 will be consumed.
1) The mass of Na2CO3 that remains after the reaction will be the initial less the mass that reacted:
m = 3.6 - 1. 6 = 2.0 g
2) All the AgNO3 reacted, so there isn't a mass present after the reaction.
m = 0 g
3) Now, doing the stoichiometry calculus between AgNO3 and Ag2CO3
2 moles of AgNO3 ------------- 1 mol of Ag2CO3
2x170 g ------------------------------- 276 g
5.14 g --------------------------------- x
By a simple direct three rule:
340x = 1418.64
x = 4.17 g of Ag2CO3
4) Now, doing the stoichiometry calculus between AgNO3 and NaNO3
2 moles of AgNO3 ----------------------- 2 moles of NaNO3
2x170 g ---------------------------------------- 2x85 g
5.14 g ------------------------------------------- x
By a simple direct three rule:
340x = 873.8
x = 2.57 g