NatalieZepeda
29.08.2019 •
Chemistry
Chemical bonds are made and broken by:
a. energy changes
c. chemical reactions
b. enzymes
d. catalysts
Solved
Show answers
More tips
- C Computers and Internet What is Web 2.0 and How Does it Work?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
Answers on questions: Chemistry
- C Chemistry How do energy and light relate to one another?...
- C Chemistry What volume in liters of 4.50 m naoh contains 7.25 mol naoh?...
- C Chemistry Why take the initial and final temperature?...
- C Chemistry 99999999999999999999999...
- C Chemistry How many mole ratios can be written for a chemical reaction involving 3 substances...
- C Chemistry 50 ml of 2m h2so4 react with 75 ml of 2m naoh. identify the limiting and excess reactants...
- C Chemistry Which sample of nitrogen gas at stp occupies the largest volume? (a) 14 liters (b) 14 moles (c) 14 grams (d) 1.4 × 10^{24} molecules (e) 4.2 × 10^{24} molecules...
- C Chemistry If the temperature and volume of a gas are both high, then what would the pressure be? lower? higher as well?...
- C Chemistry An unknown gas diffuses 0.25 times as fast as helium. what is it’s molar mass? what steps are to be done?...
- C Chemistry The atomic number of an element is equal to the number of contained in one atom of that element. protons neutrons electrons protons and neutrons the atomic number...

Ответ:
Ответ:
31.43 ml of sulfuric acid is needed to react with 23.1 g of barium peroxide.
Explanation:
For the reaction:
BaO₂(s) + H₂SO₄(aq) ⇒ BaSO₄(s) + H₂O₂(aq)
We need 1 mole of BaO₂ to react with 1 mole of H₂SO₄ and then forming 1 mole of BaSO₄ and 1 mole of H₂O₂.
We know that 4.5 M solution of H₂SO₄ means 4.5 moles of H₂SO₄ in 1000 ml of solution.
Then,
23.1 g BaO₂ ×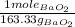 ×
×  ×
× 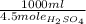 = 31.43 ml H₂SO₄
= 31.43 ml H₂SO₄