Compare the system (still at 50 °C) with a volume of 1.0 L and with 3.0 L. What is the total amount of gas (mol) present in the container at each of these volumes?
Solved
Show answers
More tips
- S Style and Beauty How to Choose the Right Fur Coat and Avoid Regrets?...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- C Chemistry The kinetic theory of matter states that gases are made of...
- C Chemistry Consider the following reaction: CO(g)+2H2(g) -- CH3OH(g). An equilibrium mixture of this reaction at a certain temperature was found to have [CO]=0.115M, [H2]=0.116M, and [CH3OH]=0.190M....
- C Chemistry Consider the reaction of magnesium metal with hydrochloric acid to produce magnesium chloride and hydrogen gas. If 3.29 mol of magnesium and 3.29 mol of hydrochloric acid are reacted,...
- C Chemistry Two people start out 100 meters apart from each other and start moving towards each other at the same time. One person is moving at half the speed of the other person and they meet...
- C Chemistry Section 5: covalent and ionic bonding bond is formed by the attraction of ions with opposite charges. covalent bond is what? ionic bond is what? bond is formed when two atoms share...
- C Chemistry What are three ways in which geologists use seismographic data?...
- C Chemistry Science grade 8 in sexual reproduction, an offspring is produced with genes from both parents. when the offspring has a new genetic variation that it did not inherit from either parent,...
- C Chemistry Which of the following challenges poses the biggest problem to the long term use of alternative fuels...
- C Chemistry A gas occupies 34.5 liters at a pressure of 345 torr what will be the volume of this gas if the pressure becomes 760 torr...
- C Chemistry Distilled vinegar contains a solution of acetic acid (CH3CO2H) in H2O. What is the concentration of the solution that results from diluting 500 ml of 0.839 M vinegar solution to 2.5...

Ответ:
The total amount of gas present in the container of 1 L is 0.0446 moles and in container of 3 L is 0.1334 moles.
Explanation:
It is known that 1 mole of any gaseous element contains 22.4 litres of that gas molecules. So in order to determine the total amount of gas present in 1 L and 3 L, we can divide the given volume with the reciprocal of 22.4 litre to determine the moles of gas at 1 L and 3 L respectively.
So for 1 L
So no. of moles in 1 L = 0.0446 moles of that gas is present in 1 L volume.
Similarly, total amount of gas present in 3 L is
No. of moles present in 3 L =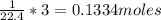
So, the total amount of gas present in 1 L is 0.0446 moles and in 3 L is 0.1334 moles. Thus, it can be stated that the number of moles increases with increase in the volume or on expansion of gas molecules.
Ответ: