Consider the following formulas and names. explain what is wrong with each name chapter 3

Solved
Show answers
More tips
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to Braid Hair with a Plaiting Machine: Tips and Recommendations...
Answers on questions: Chemistry
- C Chemistry What mass in grams would 5.7L of hydrogen gas occupy at STP?...
- C Chemistry Determine how many grams of KCL are used when 6.50 mol F2 are available F2+2KCl=2KF+Cl2...
- C Chemistry What is the enthalpy for the following reaction? overall: C + 1/2O2→CO...
- C Chemistry MAJOR HELP WILL MARK BRAINLIST!! May someone please help me answer the 18th question...
- C Chemistry How long does it take light from the sun to reach Earth? 8 minutes 20 seconds it s instant 7 years 20 days about an hour...
- C Chemistry S. De que manera han permitido estudiara los cuerpos celestes las ondasElectromagnéticas...
- P Physics What is the definition of uniform motion?...
- P Physics As a 5.0 x 10^2 newton basketball player jumps from the floor up toward the basket, the magnitude of the force of her feet on the floor is 1.0 x 10^3 newtons. as she jumps,...
- E English What does gale give to katniss after the reaping?...
- C Computers and Technology The mini toolbar contains the italic button...

Ответ:
Names ought to be:
a. Iron(II) bromide.
b. Carbon disulfide.
c. Cobalt(III) nitrate.
d. Magnesium hydroxide.
e. Copper(I) oxide.
See the explanation for why the names given are not appropriate.
ExplanationMetals and nonmetals tend to form ionic compounds when the two combine. Metals may also combine with polyatomic ions like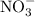 to produce an ionic compound. a, c, d, and e are all ionic compounds. Names for ionic compounds should not include numerals such as "di-" or "tri-". Also, indicate the oxidation state for transition elements using Roman numerals in brackets.
to produce an ionic compound. a, c, d, and e are all ionic compounds. Names for ionic compounds should not include numerals such as "di-" or "tri-". Also, indicate the oxidation state for transition elements using Roman numerals in brackets.
The name in a is not correct since:
Iron Fe is a transition element. Its oxidation state is missing.There's a prefix in front of "bromide" despite FeBr₂ is an ionic compound.The name in b is not correct since:
C stands for carbon. Cu is the symbol for copper. CS₂ is a covalent compound between two nonmetals. The prefix that indicates the number of sulfur atoms in the molecule is missing.The name in c is not correct since:
Cobalt Co is a transition element. Its oxidation state is missing.There's a prefix in front of "nitrate" despite Co(NO₃)₂ is an ionic compound.The name in d is not correct since:
There's a prefix in front of "hydroxide" despite Mg(OH)₂ is an ionic compound.The name in e is not correct:
There's a prefix in front of "copper" despite Cu₂O is an ionic compound.The oxidation state for copper in Cu₂O should be +1 rather than +2.Ответ:
E°(Ag⁺/Fe°) = 0.836 volt
Explanation:
3Ag⁺ + 3e⁻ => Ag°; E° = +0.800 volt
Fe° => Fe⁺³ + 3e⁻ ; E° = -0.036 volt
Fe°(s) + 3Ag⁺(aq) => Fe⁺³(aq) + 3Ag°(s) ...
E°(Ag⁺/Fe°) = E°(Ag⁺) - E°(Fe°) = 0.800v - ( -0.036v) = 0.836 volt