erichenkell2280
25.03.2020 •
Chemistry
Consider the model above. It represents the electrical force. As r increases, the attractive force decreases. How would this model
compare to a model for the force of gravity?
A)
The models would be the same. Both forces are the result of distance,
mass, and charge.
B)
There would be no way to compare the two forces as they deal with
different types of particles. Gravity is mass-based and the electrical force
is charge-based
The model of the gravitational force would be the same but without the
charges. Gravity is always attractive, but electrical force can be attractive
or repulsive.
The model of the gravitational force would vary because it does not vary
with changes in the distance between two objects. Gravity is always an
attractive force, based solely on mass.
D)
Solved
Show answers
More tips
- H Health and Medicine Want to Lose Weight? Here s How Many Calories You Need Per Day...
- S Style and Beauty How to Grow Hair Faster: Real Methods and Advice...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- B Biology Does any one know what dog breed is this?...
- M Mathematics A store sells three different styles of fleece jacket in a different color is it style can also be purchased with or without it but how many different versions of fleece...
- E English 2483422042 call mmmmmmeeeee...
- H History Este es mi novio(●♡∀♡)(●♡∀♡)(●♡∀♡)...

Ответ:
1) 0.1077 moles
2) 137043.2 moles
3) 180.186 grams
4) 6.53×10²² molecules or formula units
5) 1.505×10²³ ions
6) 90.626 grams
7) 0.555 moles
Explanation:
1) The number of moles, n = Mass of the substance/(Molar mass of the substance)
The molar weight of PO₄⁻³ is 94.971 g/mol
The number of moles in 10.23 g of PO₄⁻³ is n =10.23/94.971 = 0.1077 moles
The number of moles in 10.23 g of PO₄⁻³ = 0.1077 moles
2) The number of molecules of Na₂CO₃ in one mole of Na₂CO₃ is given by the Avogadro's number,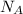 = 6.02 × 10²³ molecules
= 6.02 × 10²³ molecules
Therefore, the number of moles in 8.25 × 10²⁸ molecules of Na₂CO₃ is found by determining how many Avogadro's number of molecules are in 8.25 × 10²⁸ molecules of Na₂CO₃ as follows;
The number of moles in 8.25 × 10²⁸ molecules of Na₂CO₃ = (8.25 × 10²⁸)/(6.02 × 10²³) = 137043.2 moles
The number of moles in 8.25 × 10²⁸ molecules of Na₂CO₃ = 137043.2 moles
3) The molar mass of CH₂O = 30.031 g/mol
Therefore, the mass of 6 of CH₂O = 6 moles × 30.031 g/mol = 180.186 grams
The mass of 6 of CH₂O = 180.186 grams
4) The molar mass of NaCl = 58.44 g/mol
The number of moles of NaCl in 6.34 g of NaCl 6.34/58.44 = 0.1085 moles
1 formula unit of NaCl = 1 molecule of NaCl
The number of molecules in one mole of a substance =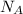 = 6.02 × 10²³
= 6.02 × 10²³
Therefore, the number of molecules in 0.1085 moles of NaCl = 6.02 × 10^(23) × 0.1085 = 6.53×10²² molecules or formula units
5) The number of ions per mole of any substance is give by Avogadro's number,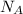 = 6.02 × 10²³ ions,
= 6.02 × 10²³ ions,
The number of ions in 0.25 moles of Cu⁺² = 0.25×6.02 × 10²³ ions = 1.505×10²³ ions
The number of ions in 0.25 moles of Cu⁺² = 1.505×10²³ ions
6) The molar mass of CH₄ = 16.04 g/mol
The number of moles, n, in 3.4×10²⁴ = 3.4×10^(24)/(6.02×10^(23)) = 5.65 moles 5.65*16.04
The mass of 5.65 moles of CH₄ = 5.65 moles × 16.04 g/mol = 90.626 grams
The mass of 3.4×10²⁴ molecules of CH₄ = 90.626 grams
7) The density of water = 1 g/mL
The volume of the water 10 mL
The mass of the water = Volume × Density = 10 mL × 1 g/mL= 10 grams
The molar mass of water = 18.015 g/mol
The number of moles of water in 10 g of water = mass/(Molar mass) = 10/18.015 = 0.555 moles.