rebecabosca
01.08.2019 •
Chemistry
Consider the reaction 2 hbr (g) à h2 (g) + br2 (g) a. express the rate of the reaction in terms of the change in concentration of each of the reactants and products. b. in the first 25.0 s of this reaction, the concentration of hbr drops from 0.600 m to 0.512 m. calculate the average rate of the reaction during this time interval.
Solved
Show answers
More tips
- A Animals and plants How ants survive winter: exploring the secrets of their winter life...
- F Food and Cooking Discover How to Properly Prepare Dough for Rasstegai...
- P Philosophy Unidentified Flying Object - What is the Nature of this Phenomenon?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- H Health Is dry oral mucosa a sign of dehydration?...
- E English 5. Which figurative language technique is used in the following sentence? I couldn t have lived through Christmas without giving you a present. I Plzz help...
- S Social Studies Describe the nature of the kkk’s attack on the catholic church. why do you suppose kkk spokespersons chose to emphasize such things in their attempts to attract...

Ответ:
2HBr(g) = H2(g)+ Br2(g)
a) Rate of reaction is the measure of the change in concentration of the reactants or the change in concentration of the product per unit time(seconds).
Therefore;
Rate = -1/2(Δ(HBr)/Δt = Δ(H₂)/Δt =Δ(Br₂)/Δt,
where ΔHBr is the change in concentration of HBr (reactant), ΔH₂ is the change in hydrogen concentration , ΔBr₂ is the change in bromine concentration and Δt is the change in time.
b) Average rate of reaction after 25 sec will be,
rate = -1/2Δ(HBr)/Δt
= -1/2 (0.512 M - 0.6 M)/(25 s-0 s)
= -1/2 (-0.088)/25
= 0.00176 M/s
Ответ:
The rate of reaction in terms of concentration change has been given by
The rate of reaction when the concentration of HBr has been dropped to 0.512 M has been 1.76 M/s.
M/s.
(a) The rate of reaction can be defined as the change in concentration of the reactant and product per unit time.
The rate of reaction in terms of reactant change can be given by:
Rate = n
where, n is the coefficient, has been the change in reactants concentration, and
has been the change in reactants concentration, and  has been the change in the time.
has been the change in the time.
For the given reaction, the rate for change in each reactant and product has been:
Rate = =
=  =
= 
(b) The change in the concentration of HBr has been:
The rate of reaction in terms of HBr concentration has been:
Rate =
Rate =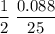
Rate = 0.00176 M/s
Rate = 1.76 M/s.
M/s.
The rate of reaction when the concentration of HBr has been dropped to 0.512 M has been 1.76 M/s.
M/s.
For more information about the rate of reaction, refer to the link:
link
Ответ:
33 grams
Explanation:
CO2 is the chemical formula of carbon dioxide.
Given that:
Amount of moles of CO2 (n) = 0.75
Mass of CO2 in grams = ?
Obtain molar mass of CO2 using the atomic masses of the individual elements
Carbon = 12g; O = 16g
CO2 = [12g + (16g x 2)
= 12g + 32g
= 44 g/mol
Since, n = mass in grams / molar mass
0.75 moles = mass in grams / 44g/mol
mass in grams = 0.75 moles x 44g/mol
mass in grams = 33
Thus, the mass of 0.75 moles of CO2 is 33 grams