xeskimopie
02.10.2019 •
Chemistry
Density i
mass
volume of metal
final water volume
step 1
use the metal’s density as a conversion factor to convert from mass (in units of grams) to volume (in units of milliliters).
step 2
the metal displaces its volume in the water so the water level rises by that volume. the final volume is determined by adding the initial volume and the volume of the metal.
you have a 100.0-ml graduated cylinder containing 50.0 ml of water. you carefully place a 151-g piece of brass (density = 8.56 g/ml) into the water. what is the final volume reading in the graduated cylinder?
ml
what is the volume that 151 g of brass occupies?
ml
if the brass displaces this volume of water, what is the final volume in the cylinder?
volume brass = 17.6 ml
ml
Solved
Show answers
More tips
- O Other What happens if you get scared half to death twice?...
- F Family and Home What s That Noise When a Kettle Boils? The Science of Water and Steam...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- W Work and Career How much does an honest traffic police officer earn in a day?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- H Health and Medicine Is Massage Necessary? Facts and Opinions...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
- F Food and Cooking When is Easter in 2011?...
Answers on questions: Chemistry
- C Chemistry Assuming the volume of the insoluble substance to be 92.5 ml and the mass to be 693.8 g, what is the density of the substance? be sure to use the correct number of...
- C Chemistry Which substance would you most likely need to cool to the lowest temperature before it condenses? a: low melting point and high boiling point b: high melting point...
- C Chemistry Plz help What is contact process??give detail?...
- C Chemistry What are the factors that affect the rate of dissolution...
- M Mathematics Can someone help me i need an answer in 20 mins...
- M Mathematics Help pls and show work...
- C Chemistry BRAINLIEST! BRAINLIEST! BRAINLIEST! Please help me with this question. John Dalton developed a simple model of the atom in 1803. Describe how and why the atomic model...
- E English In the poem Sympathy, what does the language Dunbar uses in Stanza 3-words such as beats, plea, and flings-convey to readers? the bird s sense of triumph and victory...
- E English Now you will use the diary entry that you’ve chosen and its elements that you’ve examined to develop either a one-page narrative or informative essay about the topic....
- M Mathematics By drawing a straight line on the graph, solve the equation 2xsquared -8x=x-4...

Ответ:
Final reading of the graduated cylinder = 67.64 mL
Volume occupied by the brass = 17.64 mL
final volume in the cylinder = 32.36 mL
Explanation:
Given:
Volume of graduated cylinder = 100.0 mL
Volume of water in the graduated cylinder = 50.0 mL
Mass of the piece of brass = 151 g
Density of brass = 8.56 g/mL
Now,
Volume of the brass =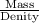
or
Volume of the brass =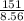
or
Volume of the brass = 17.64 mL
Hence,
final reading of the graduated cylinder = Volume of water + Volume of brass
or
the final reading of the graduated cylinder = 50 mL + 17.64 mL = 67.64 mL
Volume occupied by the brass = 17.64 mL
If the brass displaces this volume of water
then the final volume in the cylinder = Volume of water - volume of brass
= 50.00 - 17.64
= 32.36 mL
Ответ:
your answer is c) plasma
~*"auba14"*~