pedrojulianhdz2005
16.01.2020 •
Chemistry
Describe the chemical reaction of:
nh3(g)+o2(g) -> no(g)+ h2o
Solved
Show answers
More tips
- S Style and Beauty How to Properly Tie a Tie: 5 Simple Steps...
- L Leisure and Entertainment How to Make a Crab Trap in Just a Few Minutes...
- H Health and Medicine How Much Does Abortion Cost? Expert Answers and Insights...
- S Sport How to Build Arm Muscles? Effective Exercises and Tips...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
Answers on questions: Chemistry
- C Chemistry Serveral students performed this experiment without paying adequate attention to the details of the procedure. briefly explain what effect each of the following...
- C Chemistry Acertain atom has a doubly degenerate ground level pair and an upper level of four degenerate states at 450 cm−1 above the ground level. in an atomic beam study...
- C Chemistry When 0.876 g of cacl2 (110.98 g/mol) is added to 95.0 g of water at 22.4 °c in a ‘coffeecup’ calorimeter, the temperature of the resulting solution increases to...
- C Chemistry Study the picture of copper. how many connections or bonds does the center atom have to the other atoms?...
- C Chemistry The following balanced equation shows the formation of ethane (c2h6). c2h2 + 2h2 mc022-1.jpg c2h6 how many moles of hydrogen are needed to produce 13.78 mol of ethane?...
- C Chemistry What state of matter is changing from a liquid from a solid...
- C Chemistry N-14 = 95%, n-15 = 3%, n-16 = 2% c-12 = 98%, c-14 = 2%...
- C Chemistry Group-8 elements are inert and possess...
- C Chemistry What can natural selection accomplish? prove darwin s idea of survival of the fittest change relative number of inhabitants within a community favor a particular...
- C Chemistry Threonine, an amino acid whose pi is 5.6, is shown below. what would be the overall charge for threonine at a ph of 5.6?...

Ответ:
Oxidation of ammonia
Explanation:
Chemical equation:
NH₃ + O₂ → NO + H₂O
Balanced chemical equation:
4NH₃ + 5O₂ → 4NO + 6H₂O
The given reaction shows the oxidation ammonia. When oxygen react with ammonia it form nitrogen monoxide and water vapors. Nitrogen monoxide decomposes in air.
It can be seen that the given reaction also follow the law of conservation of mass. There are four nitrogen, ten oxygen and twelve hydrogen atoms on both side of equation.
According to the law of conservation mass, mass can neither be created nor destroyed in a chemical equation.
This law was given by french chemist Antoine Lavoisier in 1789. According to this law mass of reactant and mass of product must be equal, because masses are not created or destroyed in a chemical reaction.
Ответ:
Explanation:
According to the Bronsted-Lowry conjugate acid-base theory, an acid is defined as a substance which looses donates protons and thus forming conjugate base and a base is defined as a substance which accepts protons and thus forming conjugate acid.
For the given chemical equation:
Here, is loosing a proton, thus it is considered as an acid and after losing a proton, it forms
is loosing a proton, thus it is considered as an acid and after losing a proton, it forms 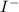 which is a conjugate base.
which is a conjugate base.
And, is gaining a proton, thus it is considered as a base and after gaining a proton, it forms
is gaining a proton, thus it is considered as a base and after gaining a proton, it forms 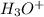 which is a conjugate acid.
which is a conjugate acid.
Hence, the correct answer is D.