makayladurham19
20.09.2019 •
Chemistry
Describe transverse waves in your own !
Solved
Show answers
More tips
Answers on questions: Chemistry
- C Chemistry Question 1 (4 points) What is the molecular weight of Magnesium nitride, Mg3N2 (OR Mg3 N2). Report your answer to two decimal places. Do not include units with...
- C Chemistry Percent yield worksheet please help and show work...
- C Chemistry g What volume of 0.10 M NaOH could be added to 200.00 mL of 0.10 M HNO2 to make an effective buffer solution...
- C Chemistry Calculate the pH of 1.00 L of the buffer 1.06 M CH3COONa/1.09 M CH3COOH before and after the addition of the following species. (Assume there is no change in volume.)...
- C Chemistry The Ka of weak acid HA is 1.0 x 10-6. A solution contains 0.25 M HA and 0.75 M KA. What is the pH of the solution after 0.17 mol HCl is added to 2.0 L of the above...
- C Chemistry Calculate the atomic mass of gallium, which is composed of two isotopes; 60.1% gallium-69, which has a mass of 68.925580 amu, and 39.9% gallium-70, which has a...
- C Chemistry Which of the following is most appropriate in measuring the mass of a cow; kilogram, gram or meter? explain your answer with detail. pl me with this one!...
- M Mathematics 12. Line ( is parallel to line m. Line 1 is a transversal with angle measures as indicated below. (3x + 42) (5x – 22) m Note: The figure is not drawn to scale....
- M Mathematics Find the unit rate. Round to the nearest hundredth, if necessary. $2.99 for 13 Ib The unit rate is $. /Ib...
- M Mathematics What is the missing number in this pattern? 0, 7, 26, 63, 124, 215, 342, __...

Ответ:
The missing nucleus is carbon-12:
Explanation:
The nuclear reaction given is:
To balance this equation you must add a nucleus which contains the missing mass and atomic numbers.
Mass number is the number of protons plus neutrons and is the superscript to the left of the chemical symbol: 249 for Cf, 257 for Rf, and 1 for n (neutron).
Atomic number is the number of protons and is the subscript to the left of the chemical symbol: 98 for Cf, 104 for RF and 0 for n.
Thus, the balance is:
Mass number:
249 + A = 257 + 4 × 1249 + A = 257 + 4249 + A = 261A = 261 - 249A = 12Atomic number:
98 + Z = 104 + 4×098 + Z = 104Z = 104 - 98Z = 6Then, you are looking for a nucleus (or gropu of nuclei) which supply 6 protons and 6 neutrons. That is a nucleus of carbon-12: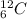 .
.
The balanced nuclear reaction is: