maltbie1283
15.04.2021 •
Chemistry
Determine how many liters of NH3 is produced if 3.6 liters of H2 reacts with an excess of N2, if all measurements are taken at the same temperature and pressure?
Solved
Show answers
More tips
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
Answers on questions: Chemistry
- B Biology Apositive benedict’s test indicates the presence of a...
- M Mathematics Vicki made 3 quarts of lemonade to serve at her party. she will serve the lemonade in glasses that hold 1 cup of liquid each. how many of these glasses can Vicki fill with lemonade?...
- M Mathematics What is m% of n please help...

Ответ:
Answer : The value of equilibrium constant (Kp) is,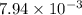
Explanation : Given,
Partial pressure of A at equilibrium = 6.70 atm
Partial pressure of B at equilibrium = 10.1 atm
Partial pressure of C at equilibrium = 3.60 atm
The given chemical reaction is:
The expression for equilibrium constant is:
Now put all the given values in this expression, we get:
Thus, the value of equilibrium constant (Kp) is,