googoomylizard
27.11.2019 •
Chemistry
Determine the energy of 2.00 mol of photons for each of the following kinds of light. (assume three significant figures.) you may want to reference (pages 299 - 308) section 7.2 while completing this problem. part a infrared radiation (1460 nm )
Solved
Show answers
More tips
- W Work and Career Which types of activities are not subject to mandatory licensing?...
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry yrup, USP, is prepared by dissolving 850 g of sucrose in sufficient purified water to make 1000 mL of syrup. Syrup has a specific gravity of 1.31. How many milliliters of...
- B Business Which of the following is a fallacy of international trade? a. trade is a zero-sum activity. b. exports increase employment in exporting industries a. import restrictions...
- E English What word is the adverb? brian and i met yesterday...
- M Mathematics Camelle manages her soccer team. She needs to order 444 practice jerseys for each player. There are yyy players....
- M Mathematics Nevaeh read 49 1/2 pages in 2 1/4 hours. If she continues reading at the same rate, how many pages will she read in an hour?...
- M Mathematics The three angles in the tiangle are 5x ,6x and 7x, what are the size of all triangle corners?...

Ответ:
The energy of 2.00 mol of photons of an infrared radiation 1460 nm is 164.08 kilo Joules.
Explanation:
where,
E = energy of a photon = ?
h = Planck's constant =
c = speed of light =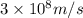
Now put all the given values in the above formula, we get the energy of the photons.
1 mol = particles
particles
Then in 2 moles of photons have:
Total energy of the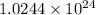 photons = E'
photons = E'
The energy of 2.00 mol of photons of an infrared radiation 1460 nm is 164.08 kilo Joules.
Ответ:
Hello! Charchar here!
has an even shorter half-life. is around a hundred times higher. Rocks from the Earth's crust have isotope ratios varying by as much as a factor of ten; this is used in geology to investigate the origin of rocks and the composition of the Earth's mantle.
Explanation:
Google!
Hope this helped!
Have a nice day!