Determine the number of moles of compound and the number of moles of each type of atom in each of the following:
(a)25.0 g of propylene, c3h6
(b) 3.06 x 10-3 g of the amino acid glycine, c2h5no2
(c)25 lb of the herbicide treflan, c13h16n2o4f (1lb =454 g)
(d)0.125 kg of the insecticide paris green, cu4(aso3)2(ch3co2)2
(e)325 mg of aspirin, c6h4(co2h)(co2ch3)
Solved
Show answers
More tips
- C Computers and Internet IMHO: What is it, why is it important, and how to use it?...
- H Health and Medicine How to Calculate Your Ideal Weight?...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- P Philosophy How to Develop Extrasensory Abilities?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- C Chemistry Plzz tell me the answer u need help im kinda confused All of the following are examples of an external stimulus except - Group of answer choices Ⓐ hunger. Ⓑ sunlight. Ⓒ a loud...
- C Chemistry I dont really know so can someone help me :) (15 points)...
- C Chemistry Actual value= -273 Experimental value= -274 Calculate the percent error of your value for absolute zero (in Celsius) Percent error = actual value - experimantal value acual...
- C Chemistry Write the formulas (example SiH4) for the following molecules: NOTE: Silicide is from silicon water Hexaboron Monosilicide Dinitrogen Trioxide Arsenic Pentabromide What type...
- C Chemistry A The Mantle is cooling down and causing the core to become solid B The mantle is getting heated by the core causing convection C The asthenosphere is heating up causing fires...
- C Chemistry When atoms join together, the bond is either covalwnt or ionic...
- C Chemistry If you have 3.54 g of h2, how many grams of nh3 can be produced?...
- M Mathematics The lengths of a fish species are normally distributed with mean 35 cm and standard deviation 8 cm. The fisheries department has decided that the smallest 10% of the fish are...
- M Mathematics Please need help due today 1: A : Suppose you are a new employee. You notice that each payment option describes a sequence and decide to use rules to help determine which option...
- H History Groups like the Ku Klux Klan resisted Reconstruction by: A. supporting the Fourteenth Amendment. B. voting for Robert Smalls. C. joining the Freedmen s Bureau. D. making life...

Ответ:
Explanation:
To calculate the number of moles, we use the equation:
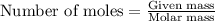 .....(1)
.....(1)
For a:Mass of propylene = 25 g
Molar mass of propylene = 42 g/mol
Putting values in equation 1, we get:
In 1 mole of propylene, 3 moles of carbon atoms and 8 moles of hydrogen atoms are present.
So, in 0.6 moles of propylene, of carbon atoms and
of carbon atoms and  of hydrogen atoms are present.
of hydrogen atoms are present.
Thus, the number of moles of compound is 0.6 moles and number of moles of carbon and hydrogen atoms are 1.8 moles and 4.8 moles respectively.
For b:Mass of glycine =
Molar mass of glycine = 75 g/mol
Putting values in equation 1, we get:
In 1 mole of glycine, 2 moles of carbon atoms, 5 moles of hydrogen atoms, 1 mole of nitrogen atom and 2 moles of oxygen atoms are present.
So, in of glycine,
of glycine,  of carbon atoms,
of carbon atoms,  of hydrogen atoms,
of hydrogen atoms,  of nitrogen atoms and
of nitrogen atoms and  of oxygen atoms are present.
of oxygen atoms are present.
Thus, the number of moles of compound is moles and number of moles of carbon, hydrogen, nitrogen and oxygen atoms are
moles and number of moles of carbon, hydrogen, nitrogen and oxygen atoms are  ,
,  ,
,  and
and  respectively.
respectively.
For c:Mass of Treflan = 25 lbs = 11350 g (Conversion factor: 1 lbs = 454 g)
Molar mass of Treflan = 283.28 g/mol
Putting values in equation 1, we get:
In 1 mole of treflan, 13 moles of carbon atoms, 16 moles of hydrogen atoms, 2 mole of nitrogen atom, 4 moles of oxygen atoms and 1 mole of fluorine are present.
So, in 40.03 moles of treflan, of carbon atoms,
of carbon atoms,  of hydrogen atoms,
of hydrogen atoms,  of nitrogen atoms,
of nitrogen atoms, 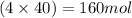 of oxygen atoms and
of oxygen atoms and  of fluorine atoms are present.
of fluorine atoms are present.
Thus, the number of moles of compound is 40 moles and number of moles of carbon, hydrogen, nitrogen, oxygen and fluorine atoms are 520 moles, 640 moles, 80 moles, 160 moles and 40 moles respectively.
For d:Mass of Paris green = 0.125 kg = 125 g (Conversion factor: 1 kg = 1000 g)
Molar mass of Paris green = 618.11 g/mol
Putting values in equation 1, we get:
In 1 mole of Paris green, 4 moles of carbon atoms, 6 moles of hydrogen atoms, 4 moles of copper atom, 10 moles of oxygen atoms and 2 moles of arsenic are present.
So, in 0.202 moles of Paris green, of carbon atoms,
of carbon atoms,  of hydrogen atoms,
of hydrogen atoms,  of copper atoms,
of copper atoms,  of oxygen atoms and
of oxygen atoms and  of arsenic atoms are present.
of arsenic atoms are present.
Thus, the number of moles of compound is 0.202 moles and number of moles of carbon, hydrogen, copper, oxygen and arsenic atoms are 0.808 moles, 1.212 moles, 0.808 moles, 2.02 moles and 0.404 moles respectively.
For e:Mass of Aspirin = 325 mg = 0.325 g (Conversion factor: 1 g = 1000 mg)
Molar mass of aspirin = 180.16 g/mol
Putting values in equation 1, we get:
In 1 mole of aspirin, 9 moles of carbon atoms, 8 moles of hydrogen atoms and 4 moles of oxygen atoms are present.
So, in 0.0018 moles of aspirin, of carbon atoms,
of carbon atoms, 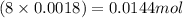 of hydrogen atoms and
of hydrogen atoms and  of oxygen atoms are present.
of oxygen atoms are present.
Thus, the number of moles of compound is 0.0018 moles and number of moles of carbon, hydrogen and oxygen are 0.0162 moles, 0.0144 moles and 0.0072 moles respectively.
Ответ:
Reduction
Oxidation
Explanation: