Determine the theoretical maximum moles of ethyl acetate, , that could be produced in this experiment. the reactant, acetic acid, is the limiting reagent. (to avoid introducing rounding errors on intermediate calculations, enter your answer to four significant figures.) theoretical maximum moles of ethyl acetate = mol reactant mass 11.0 g product mass 10.8 g reactant moles 0.1833 mol
Solved
Show answers
More tips
- L Leisure and Entertainment Choosing the Right Books to Read: Tips and Recommendations...
- L Leisure and Entertainment What can be considered an antique: defining and valuing old objects...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- C Computers and Internet Log-in: what is it and why do you need it?...
- C Computers and Internet What is the Meaning of lol and How Did it Become Popular?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- H Health and Medicine How to Help Men Gain Weight?...
- F Family and Home How to Build a Strong Relationship with Your Child: Tips for Effective Communication...
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Food and Cooking What age is appropriate for giving your child cocoa?...
Answers on questions: Chemistry
- B Biology Differential gene expression is the result of different cells containing different ....
- M Mathematics Is 8.602 × 107 greater than, less than, or equal to 8.14 × 1012 ?...
- C Computers and Technology North Hills College has decided to implement a new registration system that will allow students to register online, as well as in person. As IT manager, you decide...
- M Mathematics Unit 4: Lines Major Assessment The slope of the line that goes through (-1,5) and (3, y) is 5. Find the value of y. 7 8 9 10 11 12...
- W World Languages What is the prepositional phrase for donovan would rather have cookies before dinner...

Ответ:
0.1832 moles of ethyl acetate (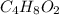 )
)
Explanation:
1. Find the balanced chemical equation:
In the production of ethyl acetate, the acetic acid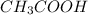 reacts with ethanol to produce ethyl acetate
reacts with ethanol to produce ethyl acetate 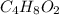 and water, that is:
and water, that is:
2. Find the theoretical maximum moles of ethyl acetate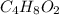 :
:
As the problem says that the acetic acid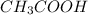 is the limiting reagent, use stoichiometry to find the moles of ethyl acetate produced:
is the limiting reagent, use stoichiometry to find the moles of ethyl acetate produced:
Ответ:
B) You are to continue heating the round-bottom flask until all liquid has been distilled.
Explanation:
Fractional distillation refers to a method that's useful for the separation of mixtures of solvents whereby there is less than 40°C in the boiling point difference.
With regards to the options given, the incorrect statement about the procedure for distillation is that you are to continue heating the round-bottom flask until all liquid has been distilled. This isn't correct.