Dinitrogen tetroxide and hydrazine (N2H4) undergo a redox reaction in which nitrogen and water are formed as products. What mass of nitrogen could be produced when 50.0 g of dinitrogen tetroxide and 45.0 g of hydrazine are combined?
Solved
Show answers
More tips
- S Society and Politics What If There s War Tomorrow, What If We Go to War?...
- S Sport How to Do a Jumping Split...
- A Animals and plants What Do Terriers Eat?...
- F Food and Cooking Discover the Benefits and Properties of Dates...
- C Computers and Internet Dynamically Assigned IP Address: What Is It and How Does It Work?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- C Chemistry I need help with these two chemistry problems I m completely stuck...
- C Chemistry Need help quick!! See picture for question...
- C Chemistry What do most 13-14 year old boys look for in a girl or how do you know if they like you?...
- C Chemistry PROJECT: KITCHEN TOOLS...
- C Chemistry A substance appears to be pure. Which of the following cannot be true. The substance is a mixture. The substance is an element. The substance is a compound. The substance has mass....
- C Chemistry When sample X is passed through a filter paper a white residue. Y. remains on the paper and a clear liquid. Z. passes through. When liquid Z is vaporized, another white residue remains....
- C Chemistry 13. Nitrogen dioxide is a gas that can be generated by emissions from vehicles and factories. It can also be generated by natural sources, such as forest fires, lightning, and microblal...
- C Chemistry What is the molecular geometry of COF2?...
- C Chemistry Are the following equations balanced or unbalanced? 1. SnO2 + 2H2 → Sn + 2 H2O 2. 4NH3 + 5O2 → 4NO + 6H2O 3. 2B2Br6 + 5HNO3 2 B(NO3)3 + HBr 4. 2KNO3 + 1H2CO3 → 1K2CO3 + 2HNO3...
- C Chemistry Which of the following types of plastic is almost never recycled? A take-out food containers B milk jugs C shampoo bottles D soda bottles...

Ответ:
The mass of nitrogen molecule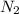 = 45.65 g
= 45.65 g
Explanation:
The equation for the redox reaction can be represented as follows:
We know that:
numbers of moles = mass/molar mass
For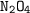 :
:
number of moles = 50g/92 g/mol
number of moles = 0.5435 mol
For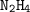 :
:
number of moles = 45 g/ 32 g/mol
number of moles = 1.40625 mol
From the above equation;
number of moles of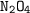 needed = 1/2 moles of
needed = 1/2 moles of 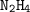 = 1/2 × 1.40625 mol
= 1/2 × 1.40625 mol
= 0.703125 mol
The amount of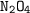 present = 0.5435 moles which is less than the needed. As such
present = 0.5435 moles which is less than the needed. As such 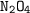 is the limiting reagent
is the limiting reagent
The number of moles of nitrogen molecule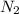 produced = 3 × (
produced = 3 × (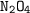 )
)
= 3 × 0.5435
= 1.6305 mol
The mass of nitrogen molecule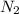 = number of moles of
= number of moles of 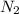 × molar mass of
× molar mass of 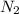
The mass of nitrogen molecule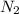 = 1.6305 mol × 28 g/mol
= 1.6305 mol × 28 g/mol
The mass of nitrogen molecule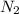 = 45.654 g
= 45.654 g
The mass of nitrogen molecule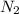 = 45.65 g
= 45.65 g
Ответ:
12.4%
Step-by-step explanation:
The results from the random sample counts 124 from a total of 1000 people that wear contact lenses. This means