tiwaribianca475
15.07.2020 •
Chemistry
Does anyone know how to do question nine if can you please show working out. thanks.
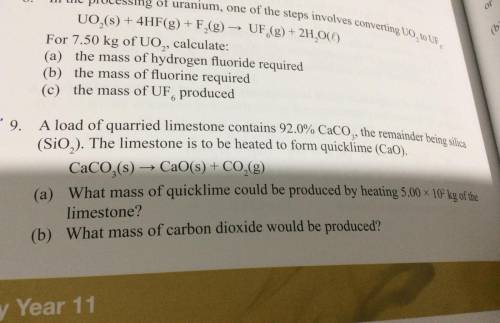
Solved
Show answers
More tips
- S Science and Technology How to Make a Homemade Smoker: The Ultimate Guide...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- M Mathematics Construct an inscribed circle in triangle PQR by finding the incenter of the triangle. PLEASE IM LITERALLY WANNA JUMP OFF A ROOF CUZ OF THIS...
- H History Why were white people who lynched black people rarely arrested?...
- M Mathematics Which of the following tables represents a proportional relationship?...

Ответ:
A. 257600g (i.e 257.6 Kg) of CaO were produced.
B. 202400g (i.e 202.4 Kg) of CO2 were produced.
Explanation:
We'll begin by calculating the mass of CaCO3 present in 5×10² Kg of lime stone. This is illustrated below:
From the question given, the lime stone contains 92% of CaCO3.
Mass of CaCO3 = 92% x 5×10² Kg
Mass of CaCO3 = 92/100 x 5×10² Kg
Mass of CaCO3 = 460 Kg
Therefore, 460 kg of CaCO3 is present in the lime stone.
Next, we shall determine the mass of CaCO3 that was heated and the masses of CaO and CO2 produced from the balanced equation. This is illustrated below:
CaCO3(s) —> CaO(s) + CO2(g)
Molar mass of CaCO3 = 40 + 12 + (16x3) = 100g/mol
Mass of CaCO3 from the balanced equation = 1 x 100 = 100g
Molar mass of CaO = 40 + 16 = 56g
Mass of CaO from the balanced equation = 1 x 56 = 56g
Molar mass of CO2 = 12 + (2x16) = 44g/mol
Mass of CO2 from the balanced equation = 1 x 44 = 44g
Summary:
From the balanced equation above,
100g of CaCO3 were heated to produce 56g of CaO and 44g of CO2.
A. Determination of the mass of quick lime, CaO produced by heating 5×10² Kg of lime stone.
5×10² Kg of lime stone contains 460 Kg (i.e 460×10³ g) of CaCO3.
From the balanced equation above,
100g of CaCO3 were heated to produce 56g of CaO.
Therefore, 460×10³g of CaCO3 will be heated to produce = (460×10³ x 56)/100 = 257600g of CaO.
Therefore, 257600g (i.e 257.6 Kg) of CaO were produced.
B. Determination of mass of carbon dioxide, CO2 produced by heating 5×10² Kg of lime stone.
5×10² Kg of lime stone contains 460 Kg (i.e 460×10³ g) of CaCO3.
From the balanced equation above,
100g of CaCO3 were heated to produce 44g of CO2.
Therefore, Therefore, 460×10³g of CaCO3 will be heated to produce = (460×10³ x 44)/100 = 202400g of CO2.
Therefore, 202400g (i.e 202.4 Kg) of CO2 produced.
Ответ:
eee
Explanation: