brutusjohnson1
22.12.2020 •
Chemistry
DOG SKELETAL SYSTEM BONES SKELETAL TISSUE BONE CELLS SMALLEST TO LARGEST
Solved
Show answers
More tips
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- S Style and Beauty How to braid friendship bracelets?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- C Computers and Internet How to Learn to Type Fast?...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
Answers on questions: Chemistry
- C Chemistry What is the mass of 2.86 moles of silver (Ag)?...
- C Chemistry All of the following are examples of allotropes of carbon EXCEPT Choose the one alternative that best completes the statement or answers the question. Choose the one alternative that...
- M Mathematics What are 2 numbers that make 49, i need with my math homework. i’m in 3rd grade...
- H History Atlanta mayor kasim reed has been a major supporter of the plan to deepen the savannah seaport. when funding was approved for the project, reed stated, work can now begin in earnest...
- M Mathematics The average cost of pet supplies is $66 per month and has a standard deviation of $21. Make a statement comparing a family who spends $45 per month on pet supplies....
- H History The central idea of this paragraph is that Albo Read this paragraph from The Dark Game. To assure that the intelligence reached the commanders as quickly as possible--the postal services...

Ответ:
tissue bone cells ,bones skeletal,dog skeletal system
Explanation:
Ответ:
The empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.
Explanation:
The chemical equation for the combustion of hydrocarbon having carbon and hydrogen follows:
where, 'x' and 'y' are the subscripts of Carbon, hydrogen and oxygen respectively.
We are given:
Mass of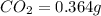
Mass of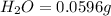
We know that:
Molar mass of carbon dioxide = 44 g/mol
Molar mass of water = 18 g/mol
For calculating the mass of carbon:
In 44 g of carbon dioxide, 12 g of carbon is contained.
So, in 0.364 g of carbon dioxide, of carbon will be contained.
of carbon will be contained.
For calculating the mass of hydrogen:
In 18 g of water, 2 g of hydrogen is contained.
So, in 0.0596 g of water,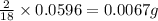 of hydrogen will be contained.
of hydrogen will be contained.
To formulate the empirical formula, we need to follow some steps:
Step 1: Converting the given masses into moles.Moles of Carbon =
Moles of Hydrogen =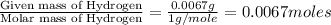 ]
]
Step 2: Calculating the mole ratio of the given elements.For the mole ratio, we divide each value of the moles by the smallest number of moles calculated which is 0.0067 moles.
For Carbon =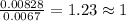
For Hydrogen =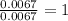
Step 3: Taking the mole ratio as their subscripts.The ratio of C : H = 1 : 1
The empirical formula for the given compound is CH
For determining the molecular formula, we need to determine the valency which is multiplied by each element to get the molecular formula.
The equation used to calculate the valency is:
We are given:
Mass of molecular formula = 128.2 g/mol
Mass of empirical formula = 13 g/mol
Putting values in above equation, we get:
Multiplying this valency by the subscript of every element of empirical formula, we get:
The general formula of an alkane is , where n = any natural number
, where n = any natural number
Here, n = 10 and it does not satisfy being an alkane
Hence, the empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.