el mercurio es un objeto metalico muy pesado en aleaseones con otro metales como oro y plata se conose cono amalgama q fenoneno es
Solved
Show answers
More tips
- F Food and Cooking How to Cook a Steak with Mustard Sauce? What Kind of Meat to Choose?...
- P Photography and Videography What is lens calibration and why is it needed?...
- F Family and Home Stay Warm but Don t Overheat: What is the Optimal Temperature for Your Home During Winter?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- C Chemistry The photo above shows a landform surrounded by water on 3 sides. we can describe this landform as an...
- C Chemistry Use the drop-down menus to classify the elements as alkali metals, alkaline earth metals, or transition metals. nickel (ni): potassium (k): cadmium (cd): calcium...
- C Chemistry Which of the following blackbody curves is representative of stars like our Sun? C A B...
- C Chemistry Tách nước từ 14,8g ancol thu được 11,2g anken. CTPT của ancol là...
- C Chemistry What are protons and neutrons...
- C Chemistry Precipitation reactions: hg(no3)2(aq) +na2cro4(aq) = (aq) + nano3 (s)...
- C Chemistry If a dna molecule is found to be made of 40% thymine,what percentage guanine would you expect...
- C Chemistry Some one pls help ill give you brainlist to the best answer....
- C Chemistry What is the molarity in .650 L of solution containing 1.08 moles of sodium chloride?...
- C Chemistry if 120.17 g of solid silicon dioxide react with 72.1g of soils mono-atomic carbon and form the products measuring 80.193 g of silicon carbide what if the predicted...

Ответ:
Answer : The percentage mass of excess with respect to initial mass is 60.0 %
Solution : Given,
Mass of = 40 g
= 40 g
Mass of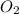 = 25 g
= 25 g
Molar mass of = 64 g/mole
= 64 g/mole
Molar mass of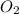 = 32 g/mole
= 32 g/mole
First we have to calculate the moles of and
and 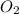 .
.
Now we have to calculate the limiting and excess reagent.
The balanced chemical reaction is,
From the balanced reaction we conclude that
As, 2 mole of react with 1 mole of
react with 1 mole of 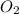
So, 0.625 moles of react with
react with  moles of
moles of 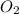
From this we conclude that,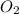 is an excess reagent because the given moles are greater than the required moles and
is an excess reagent because the given moles are greater than the required moles and  is a limiting reagent and it limits the formation of product.
is a limiting reagent and it limits the formation of product.
Now we have to calculate the excess moles of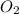
Excess moles of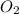 = Required moles - Given moles
= Required moles - Given moles
Excess moles of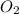 = 0.781 - 0.312 = 0.469 mole
= 0.781 - 0.312 = 0.469 mole
Now we have to calculate the excess mass of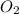
Excess mass of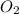 = 15.0 g
= 15.0 g
Initial mass of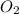 = 25 g
= 25 g
Now we have to calculate the percentage mass of excess with respect to initial mass.
Therefore, the percentage mass of excess with respect to initial mass is 60.0 %