Enter your answer in the provided box. a sample of sulfur hexafluoride gas occupies 9.53 l at 215°c. assuming that the pressure remains constant, what temperature (in °c) is needed to reduce the volume to 4.36 l? report your answer to the proper number of significant figures.
Solved
Show answers
More tips
- A Auto and Moto Discovering the Leader: What is the Most Expensive Car in the World?...
- H Health and Medicine Hangover: How to Get Rid of It Quickly?...
- S Style and Beauty How to Choose the Right Fur Coat and Avoid Regrets?...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- H History World history term for payment made from a lesser group to a greater one to preserve itself...
- H History Platón y Aristóteles no se ocuparon de la política...
- S Spanish Spanish IV Don José, creo que usted (sufrir) de mucho estrés. Usted (trabajar) demasiado y no (cuidarse) lo suficiente. Es necesario que usted (dormir) más horas. No creo que usted...
- M Mathematics Since obtaining measurements of the human brain is difficult with living subjects, Gladstone (1905) used cadavers to investigate if there was a relationship between a subject s head...

Ответ:
Explanation:
According to ideal gas equation, the product of pressure and volume equals to the product of number of moles, gas constant and temperature.
Mathematically, PV = nRT
Since, it is given that pressure is constant and Charle's law states that at constant pressure volume of an ideal gas is directly proportional to the absolute temperature.
Therefore,
So, number of moles will also be constant then. Hence, the formula will be as follows.
As it is given that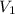 is 9.53 L,
is 9.53 L, 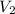 is 4.36 L,
is 4.36 L, 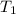 is
is 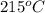 that is also equal to (215 + 273) K = 488 K.
that is also equal to (215 + 273) K = 488 K.
Now, putting these values into the formula as follows.
Temperature in degree celsius will be (223.26 K - 273 K) =
Thus, we can conclude that temperature (in °C) is needed to reduce the volume to 4.36 L is .
.
Ответ: