Explain how you would make up a one molar (1m) solution of ethyl alcohol.
Solved
Show answers
More tips
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- F Food and Cooking How to Calculate the Gender of Your Child with Blood?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- E English At the beginning of ch. 3, Holden says, I’m the most terrific liar you ever saw in your life. It’s awful. If I’m on my way to the store to buy a magazine, even, and somebody asks...
- C Chemistry The temperature of the cooling water as it leaves the hot engineof a certain automobile is 244°f. after it passes through theradiator it has a temperature of 177°f. calculate the...

Ответ:
The formula of molarity is:
Number of moles is given as:
So, the formula of molarity can be written as:
Molar mass of ethyl alcohol, =
= 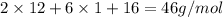
Molarity of ethyl alcohol, = 1 molar (given)
= 1 molar (given)
Let the volume of solvent that is water be 1 L.
Substituting the values in formula (1):
Since, ethyl alcohol is liquid in room temperature:
Density of ethyl alcohol is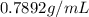
Density =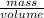
So, volume of ethyl alcohol =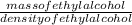
Volume of ethyl alcohol =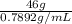
Volume of ethyl alcohol =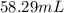
Hence, by dissolving 58.29 mL of ethyl alcohol in 1 L of water, 1 M solution of ethyl alcohol can be prepared.
Ответ:
3
2
1
4
6
5
not a 100% sure but i think this is it.