ChefCurtis
12.11.2019 •
Chemistry
For all of the following questions 20.00 ml of 0.200 m hbr is titrated with 0.200 m koh.
region 1: initial ph: before any titrant is added to our starting material
what is the concentration of h+ at this point in the titration?
m
what is the ph based on this h+ ion concentration?
Solved
Show answers
More tips
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine Reasons for the Appearance of Warts: Everything You Need to Know...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
Answers on questions: Chemistry
- C Chemistry Hi Juan, when you submit this form, the owner will be able to see your name and email address. 1 The table shows the physical properties of four elements. Which element...
- H Health Advertisers use what technique to get you to buy their products and services...
- E English Which sentence most clearly describes a plot, as opposed to just a narrative? A) A boy grows up in a Jewish neighborhood in New York City in the 1950s. B) A dancer proves...
- A Arts What does the principle of unity help achieve in a design? O A It establishes the direction of view of the items in a design composition. B. It helps group similar items...
- M Mathematics NO LINKS OR ANSWERING QUESTIONS YOU DON T KNOW The point (-2, 5) is reflected over an axis and the new point is located in Quadrant 3.a. Describe the reflection that...

Ответ:
Answer :
The concentration of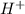 before any titrant added to our starting material is 0.200 M.
before any titrant added to our starting material is 0.200 M.
The pH based on this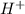 ion concentration is 0.698
ion concentration is 0.698
Explanation :
First we have to calculate the concentration of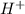 before any titrant is added to our starting material.
before any titrant is added to our starting material.
As we are given:
Concentration of HBr = 0.200 M
As we know that the HBr is a strong acid that dissociates complete to give hydrogen ion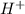 and bromide ion
and bromide ion 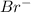 .
.
As, 1 M of HBr dissociates to give 1 M of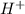
So, 0.200 M of HBr dissociates to give 0.200 M of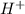
Thus, the concentration of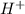 before any titrant added to our starting material is 0.200 M.
before any titrant added to our starting material is 0.200 M.
Now we have to calculate the pH based on this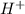 ion concentration.
ion concentration.
pH : It is defined as the negative logarithm of hydrogen ion concentration.
Thus, the pH based on this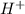 ion concentration is 0.698
ion concentration is 0.698
Ответ:
Pure substances are made up of a bulk quantity of individual atoms or molecules.
Each pure substance is made up of one of Individual atoms of the same type that are connected to form extended structures.
A chemical reaction happens when substances break apart or combine to form one or more new substances.
New substances form when bonds break and new bonds form.