isaiah1027
21.02.2020 •
Chemistry
For the following reaction, Kc = 255 at 1000 K. CO(g)+Cl2(g)⇌COCl2(g) A reaction mixture initially contains a CO concentration of 0.1550 M and a Cl2 concentration of 0.172 M at 1000 K.
What is the equilibrium concentration of CO at 1000 K?
What is the equilibrium concentration of Cl2 at 1000 K?
What is the equilibrium concentration of COCl2 at 1000 K?
Solved
Show answers
More tips
- H Health and Medicine Why do our Joints Crack?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry Me podrian ayudar a rresponde...
- C Chemistry I HAVE 25 MINUTES TO FINISH True or false the deeper you go into Earth the temperature goes up ?...
- C Chemistry Explain difference in melting points for the elements Na and Mg...
- C Chemistry Free points i love you say it back...
- C Chemistry A 2 kg medicine ball is thrown with an acceleration of 3 m/sec^2 towards the wall. With what force will it hit the wall? can someone please answer...
- C Chemistry What principle states that the volumes of gases that combine in a chemical reaction are in the ratio of small whole numbers...
- C Chemistry The rock cycle can be thought of as a “journey” that one type of rock goes through to become a different type of rock. Compare the “journeys” of an intrusive igneous rock...
- C Chemistry How would i work out a question like this ?...
- C Chemistry A flask containing neon gas is connected to an open-ended mercury manometer. The open end is exposed to the atmosphere, where the barometric pressure is 734. torr. The...
- C Chemistry Why is a pencil not a system...

Ответ:
Given reaction is:
0.1550 0.172 O initial
initial
0.1550-x 0.172-x x Equilibrium
Equilibrium
Now,
→![K_c = \frac{[COCl_2]}{[CO][Cl_2]}](/tpl/images/0519/6554/f9140.png)
By substituting the values, we get
or,
(a)
Equilibrium constant of [CO],
=
=
(b)
Equilibrium constant of [Cl₂],
=
=
(c)
Equilibrium constant of [COCl₂],
=
=
Thus the above responses are correct.
Learn more about equilibrium here:
link
Ответ:
The equilibrium concentration of CO at 1000 K is 0.016 M , the equilibrium concentration of at 1000 K is= 0.033 M and the equilibrium concentration of
at 1000 K is= 0.033 M and the equilibrium concentration of  at 1000 K is 0.139 M
at 1000 K is 0.139 M
Explanation:
Initial concentration of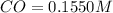
Initial concentration of
The given balanced equilibrium reaction is,
Initial conc. 0.1550 M 0.172 M 0 M
At eqm. conc. (0.1550-x) M (0.172-x) M (x) M
The expression for equilibrium constant for this reaction will be,
we are given :
Now put all the given values in this expression, we get :
Thus the equilibrium concentration of CO at 1000 K is= (0.1550-x) M =(0.1550-0.139) M = 0.016 M
Thus the equilibrium concentration of at 1000 K is= (0.172-x) M =(0.172-0.139) M = 0.033 M
at 1000 K is= (0.172-x) M =(0.172-0.139) M = 0.033 M
Thus the equilibrium concentration of at 1000 K is= x M = 0.139 M
at 1000 K is= x M = 0.139 M
Ответ:
B
Explanation: