tamiawilliams3pe55hs
03.08.2019 •
Chemistry
For the reaction 4fecl2(aq) + 3o2(g) → 2fe2o3(s) + 4cl2(g), what volume of a 0.945 m solution of fecl2 is required to react completely with 4.32 ×1021 molecules of o2?
Solved
Show answers
More tips
- A Animals and plants How ants survive winter: exploring the secrets of their winter life...
- F Food and Cooking Discover How to Properly Prepare Dough for Rasstegai...
- P Philosophy Unidentified Flying Object - What is the Nature of this Phenomenon?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- E English I want to write a book. Can some people give me some ideas of what to write about?...
- E English Which of the following sequences is the clearest example of a single cause with multiple effects, as presented in Lessons of Dr. Martin Luther King, Jr. ? h O A Farm...
- M Mathematics Aman works five consecutive days for 8 hours 7 and 1/2 hours 8 1/4 hours 6 and 1/2 hours 6 and 3/4 hours how much money will he earn for the 5 days if he makes $15.50...
- B Biology Why do animals breathe more heavily when exercising?...

Ответ:
0.01M
Explanation:
Given paramters:
Volume of FeCl₂ = ?
Concentration = 0.945M
Number of molecules of O₂ = 4.32 x 10²¹molecules
Solution:
The balanced reaction equation is given below:
4FeCl₂ + 3O₂ → 2Fe₂O₃ + 4Cl₂
Now, to solve the problem we use mole relationship between the compounds.
We work from the known to the unknown compounds. Here, the known is the oxygen gas because from the given parameters we can estimate the number of moles of the reacting gas.
number of moles of O₂ =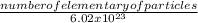
number of moles of O₂ =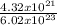
number of moles of O₂ = 0.007mole
now, from the reaction equation we find the number of moles of the FeCl₂:
3 mole of O₂ reacted with 4 moles of FeCl₂
0.007mole of O₂ reacted with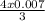 = 0.0096mole O₂
= 0.0096mole O₂
Now to find the volume of FeCl₂ we use the expression below;
volume of FeCl₂ =
=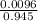
= 0.01L
Ответ:
However, when she looks at a sample using a magnifying lens, she can see small droplets of liquid surrounded by another liquid. Which conclusion best fits her observations? The mixture is a solution because it has the same appearance throughout the sample
pls mark me the brainliest