HealTheWorld
05.05.2020 •
Chemistry
For the reaction shown, calculate how many moles of NH3 form when 16.72 moles of reactant completely reacts:
3 N2H4 (l)
⟶
4 NH3(g) + N2(g)
Solved
Show answers
More tips
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- S Sport How to Do a Jumping Split...
- H Health and Medicine How Did Inna Lose Weight on Dom 2?...
- F Family and Home How to Properly Fold Napkins in a Napkin Holder?...
- F Food and Cooking How to Set Up Ventrilo - The Ultimate Guide...
- S Science and Technology How to Make a Homemade Smoker: The Ultimate Guide...
- A Auto and Moto Battle for the Relocation of The Cherkizovsky Market: Who Won?...
- C Computers and Internet How Do You Refill Cartridges?...
- S Style and Beauty Tricks and Tips: How to Get Rid of Freckles...
Answers on questions: Chemistry
- C Chemistry Choose one food chain in the web. Name all of the organisms in that chain. Start with a producer and end with the top level consumer....
- B Business Works and duties of ANM...
- B Biology Plate motion lesson 4.3 activity 1. Argument. Running puts more pressure on the knee joint and makes muscles work harder than walking does. Hel...
- L Law Why is the media sometimes called the 4th branch of government?...

Ответ:
Answer : The moles of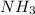 formed are, 22.3 moles.
formed are, 22.3 moles.
Explanation : Given,
Moles of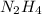 = 16.72 mol
= 16.72 mol
The given chemical reaction is:
From the balanced chemical reaction, we conclude that:
As, 3 moles of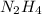 react to give 4 moles of
react to give 4 moles of 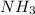
So, 16.72 moles of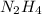 react to give
react to give 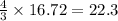 moles of
moles of 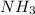
Therefore, the moles of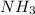 formed are, 22.3 moles.
formed are, 22.3 moles.
Ответ:
Balanced chemical reaction (dissociation):
Ba₃(PO₄)₂(s) → 3Ba²⁺(aq) + 2PO₄³⁻(aq).
[Ba²⁺] = 0,0005 M.
Ksp(Ba₃(PO₄)₂) = 3,4·10⁻²³.
Ksp = [Ba²⁺]³ · [PO₄³⁻]².
3,4·10⁻²³ = (0,0005 M)³ · [PO₄³⁻]².
[PO₄³⁻] = √3,4·10⁻²³ ÷ 1,25·10⁻¹⁰.
[PO₄³⁻] = 5,2·10⁻⁷ M.
Ksp is the solubility product constant for a solid substance dissolving in an aqueous solution.