jamilecalderonpalaci
10.12.2021 •
Chemistry
Fossils are the preserved remains or traces of an organism that used to be alive.

Solved
Show answers
More tips
- S Society and Politics Why are thugs called gopniks ? A fascinating journey through Russian subculture...
- A Animals and plants Want a Perfect Lawn? Learn How to Plant Grass the Right Way...
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
Answers on questions: Chemistry
- C Chemistry Aliquid is placed in an evacuated, sealed flask. when will this closed system reach dynamic equilibrium? when all of the liquid has evaporated to form a vapor when...
- M Mathematics a mechanic has the following drills bits 1/2 7/16 3/8 17/32 which shows the drills bits in order form smallest to largest...
- M Mathematics Enter the solution to the equation in the box. 5(x−8)=7(x−4) x =...
- S Spanish Read and choose the correct option of the verb in the correct form and tense. En la niñez, mi hermana y yo de nuestros personajes favoritos por las noches. habla...
- E English The category 1 to 5 rating known as the saffir-simpson hurricane scale provides an estimate of a hurricane’s potential to blow away small buildings, completely destroy...

Ответ:
pH of the solution is 3.70
Explanation:
The given weak acid (HA) and it's sodium salt (NaA) forms a buffer system. According to Henderson-Hasselbalch equation for this buffer system-
Where, and
and  represent number of moles of
represent number of moles of 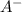 ion and HA respectively.
ion and HA respectively.
Here and
and 
So,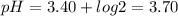
Hence pH of the solution is 3.70