clarkster112679
15.07.2020 •
Chemistry
g Calculate the energy of a photon that has a wavelength of 965 nm. 2.06x10-33 J 2.06x10-19 J 3.78x10-28 J 3.78x10-29 J None of these
Solved
Show answers
More tips
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
Answers on questions: Chemistry
- E English Whoever writes me a good (i mean good) poem, gets brainliest and (any trolls will get reported)....
- H Health You see a buoy with both red and green bands. what does this buoy mark?...
- E English Will mark as brainliesttwo teams practiced basketball in the gym. they took a break to drink water. both of the teams basketballs were left in the gym. question: what is the...
- M Mathematics Use the protractor to measure these angles M m m Check...

Ответ:
The energy of a photon that has a wavelength of 965 nm is
Explanation:
The relation between energy and wavelength of light is given by Planck's equation, which is:
where,
E = energy of the photon =
h = Planck's constant =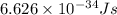
c = speed of light =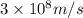
Putting in the values we get:
Thus the energy of a photon that has a wavelength of 965 nm is
Ответ: