annapittbull12
16.04.2021 •
Chemistry
g Consider (12.5 A) micro-grams of a radioactive isotope with a mass number of (78 B) and a half-life of (32.6 C) million years. If energy released in each decay is 32.6 keV, determine the total energy released in joules (J) in 1 (one) year. Give your answer with three significant figures.
Solved
Show answers
More tips
- A Animals and plants Want a Perfect Lawn? Learn How to Plant Grass the Right Way...
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...

Ответ:
Energy released = 18.985 J
Explanation:
The exponential decay of radioactive substance is given by -
N(t) = N₀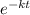
where
N₀ = initial quantity
k = decay constant
Half life,
⇒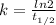
Given,
N₀ = 12.5 + 3 = 15.5 × 10⁻⁶ gm
So,
Now,
N(1) = 15.5 × 10⁻⁶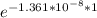
= 15.49999978904
Now,
Substance decayed = N₀ - N(t)
= 15.5 × 10⁻⁶ - 15.49999978904 × 10⁻⁶
= 21.095 × 10⁻¹⁷ kg
⇒Δm = 21.095 × 10⁻¹⁷ kg
So,
Energy released = Δmc²
= 21.095 × 10⁻¹⁷ × 3 ×10⁸ × 3 × 10⁸
= 189.855 ×10⁻¹
= 18.985 J
⇒Energy released = 18.985 J
Ответ:
The given question is incomplete. The complete question is as follows.
On the basis of periodic trends, choose the larger atom from each pair (if possible): match the elements in the left column to the appropriate blanks in the sentences on the right. make certain each sentence is complete before submitting your answer.
Sn, F, Ge, not predictable, Cr
Of Ge or Po, the larger atom is
Of F or Se, the larger atom is
Of Sn or I, the larger atom is
Of Cr or W, the larger atom is
Explanation:
When we move down a group then there occurs an increase in atomic size of the atoms due to increase in the number of electrons.
Ge is a group 14 element which lies in period 4 and Po is a group 16 element that lies in period 6. As polonium is larger in size as compared to germanium.
Fluorine is a group 17 element and lies in period 2. Selenium is a group 16 element lies in 4. Therefore, selenium is larger in size as compared to fluorine.
Sn is a group 14 element that lies in period 5 and I is a group 17 element that lies in period 5. Hence, I is a larger atom.
Cr is a d-block element which lies in period 4 and W is also a d-block element which lies in period 6. Hence, W is larger in size than Cr.
Thus, we can conclude that given blanks are matched as follows.
Of Ge or Po, the larger atom is Po.Of F or Se, the larger atom is Se.Of Sn or I, the larger atom is I.Of Cr or W, the larger atom is W.