finnthedino
14.12.2019 •
Chemistry
Give the approximate bond angle for a molecule with a linear shape.
Solved
Show answers
More tips
- D Dating, Love, Relationships How Long Can Love Last?...
- S Science and Technology How to Make a Homemade Smoker: The Ultimate Guide...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry What is environment?write in a sentence ...
- C Chemistry The label 2‑ethyl‑2,4,6‑trimethylheptane was found on a bottle of liquid hydrocarbons. although the name defines a structure unambiguously, it is not the correct iupac substitutive...
- M Mathematics I bet nobody will anser this =P. Solve 52.425 divided by 0.5...
- H Health Which of these belong at the top of the activity pyramid? a.jogging b.cardio work-outs c.video games d.yoga...

Ответ:
The approximate bond angle for a molecule with a linear shape is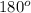
Explanation:
Linear shape is the basic shape of the organic molecules. All diatomic molecules has linear shape. These two atoms are bonded together by either single,double or triple bond.
For example, HCl has singe bond.
Some tri atomic molecules are also be linear.For example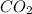 .
.
In the shape of carbondioxide the central atom is carbon and it hare four valence electrons to the each oxygen atom.And make double bond with each oxygen atom.
The shape of the molecule is linear and bond angle is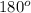 .
.
Ответ:
A) Colonists should not have to pay taxes to the British.