Given 8.10g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield?
Solved
Show answers
More tips
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- C Chemistry what effect can the presence of microscopic organisms or mud have on the color of water that is observed?...
- C Chemistry If you start with 36 hydrogen (Hz) molecules, how many nitrogen (N2) molecules will you need?...
- C Chemistry What is the top quality you were looking for in assessing a material intended to hold a molten metal? A. Low thermal conductivity B. High thermal conductivity C. High...
- C Chemistry Why is it impossible for parents with the genes Bb and BB for hair color to have a child with brown hair?...
- C Chemistry Which statement is true of convex mirrors? a. they can form real or imaginary images b. they can only form imaginary images c. they can form either virtual or real images...
- C Chemistry Photosynthesis is a vital process for plants, animals and humans, and the environment? give at least two examples of how plants, animals and humans, and the environment...
- C Chemistry I. define ionic bonding and describe how it can be formed between sodium and chlorine ii. state 3 properties of the compoundIII. what type of bonds is formed between two...
- M Mathematics What is the explicit function that defines this sequence? - 4,11,18,25...
- M Mathematics Holly, the author, has written 160 pages of her next book. She needs to write a minimum of 20 pages per day to complete the expected 380 page book. Which inequality below...
- M Mathematics PLZ help I will mark as brainliest Decode the addition or subtraction problem (Change asterisks to digits, to make the operation correct.) 6*5* − *8*4=2856...

Ответ:
10.7 g of ethyl butyrate would be synthesized in 100 % conversion
Explanation:
1 mol of butanoic acid produces 1 mol of ethyl butyrate in 100% conversion.
Number of moles = (mass/molar mass)
Molar mass of butanoic acid = 88.11 g/mol
Molar mass of ethyl butyrate = 116.16 g/mol
So, 8.10 g of butanoic acid = butanoic acid = 0.09193 moles of butanoic acid
butanoic acid = 0.09193 moles of butanoic acid
So, in 100 % conversion, moles of ethyl butyrate would be produced = 0.09193 moles
So, in 100 % conversion, mass of ethyl butyrate would be produced = = 10.7 g
= 10.7 g
Ответ:
Further Explanation:
Stoichiometry:
It is used to determine the amount of species present in the reaction by the relationship between reactants and products. It is used to determine the moles of a chemical species when moles of other chemical species present in the reaction is given.
Consider the general reaction,
Here,
A is the reactant.
B is the reactant.
C is the product.
One mole of A reacts with two moles of B to produce three moles of C. The stoichiometric ratio between A and B is 1:2, the stoichiometric ratio between A and C is 1:3 and the stoichiometric ratio between B and C is 2:3.
The given reaction occurs as follows:
The formula to calculate the moles of is as follows:
is as follows:
Substitute 8.10 g for the given mass of and 88.11 g/mol for the molar mass of
and 88.11 g/mol for the molar mass of  in equation (1).
in equation (1).
According to the stoichiometry of the reaction, one mole of reacts with one mole of
reacts with one mole of 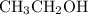 to form one mole of
to form one mole of  and one mole of
and one mole of  . So the number of moles of
. So the number of moles of  produced by 0.0919 moles of are also 0.0919 mol.
produced by 0.0919 moles of are also 0.0919 mol.
The formula to calculate the mass of is as follows:
is as follows:
Substitute 0.0919 mol for the moles of and 116.16 g/mol for the molar mass of
and 116.16 g/mol for the molar mass of  in equation (2).
in equation (2).
Since the yield is 100 %, so the amount of ethyl butyrate produced is 10.675 g.
Learn more:
1. Calculate the moles of chlorine in 8 moles of carbon tetrachloride:
2. Calculate the moles of ions in the solution:
Answer details:
Grade: Senior School
Subject: Chemistry
Chapter: Mole concept
Keywords: stoichiometry, ethyl butyrate, 0.0919 mol, one mole, 116.16 g/mol, 88.11 g/mol, 10.675 g.
Ответ:
2.998*10^8m/s 1hr K H D B D C M
=4996666.67 m/hr = 4996.67km/hr
1 60sec.