hmskevinjacobo5471
06.09.2019 •
Chemistry
Gold has an atomic weight of 196.97 g/mol, and a density of 19.3 g/cm3. approximately how many atoms are in a spherical gold nanoparticle 10 nm in diameter?
Solved
Show answers
More tips
- C Computers and Internet Make Money Online: Secrets and Essential Ways...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- C Computers and Internet How to Learn to Type Fast?...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- F Food and Cooking How to Make Delicious Cabbage Pies: The Best Recipes!...
Answers on questions: Chemistry
- C Chemistry Mary pulls a 236 gram cup (c = 0.84 J/g°C) out of the cabinet that was sitting at room temperature of 22°C. She pours a can of Sprite she got out of the refrigerator (5°C) and...
- C Chemistry la ecuación química que representa la siguiente descripción de una reacción química: Un átomo de hierro (Fe) en estado sólido reacciona con dos moléculas de ácido clohídrico (HCl)...
- C Chemistry Is the direction of daily drift different from the direction over a longer time scale? Help pls!...
- C Chemistry Whenever there is a change of state, such as a solid to liquid or liquid to gas, heat energy can be added without a temperature change because a) the added energy is not needed....
- C Chemistry An aqueous solution of potassium carbonate combine with a solution of calcium nitrate. what are the total and net ionic equation?...
- C Chemistry What does it mean to say we understand many chemical reactions as transfers of electrons...
- C Chemistry Which fraction contains the largest molecules? A) diesel B)gasoline C)kerosene?...
- C Chemistry . Give an example of a salt that is less soluble when the temperature increases...
- C Chemistry Ibg-ecrw-ntwhere come girls...
- C Chemistry M A solution is prepared by dissulving 4.3829 g of g barium hydroxide in 500.0 mL volumetric flask and diluting it to the calibration mark. Calculate the: Ba(OH)2 i) Normality...

Ответ:
The number of atoms in spherical gold nano particle are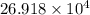
Explanation:
To calculate the volume of sphere, we use the equation:where,
r = radius of the sphere = (Conversion factor:
(Conversion factor:  )
)
Putting values in above equation, we get:

To calculate mass of of the substance, we use the equation:Volume of gold =
Density of gold =
Putting values in above equation, we get:

To calculate the number of moles, we use the equation:Mass of gold =
Molar mass of gold = 196.97 g/mol
Putting values in above equation, we get:
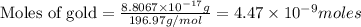
According to mole concept:1 mole of an element contains number of atom
number of atom
So, moles of gold will contain =
moles of gold will contain =  number of atoms.
number of atoms.
Hence, the number of atoms in spherical gold nano particle are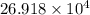
Ответ:
The correct answer is transfer pipette that had markings every 0.1 mL
Explanation:
Flask, beaker , pipette and graduated cylinder are the apparatus that are used in the lab to measure the volume of a chemical.
For an experiment, the measurement of chemical used must be precise and for that purpose, pipette is used.
All the other apparatus does not give precise measurements and hence cannot be used for measuring a chemical that has to be used in an experiment.
Hence, the correct answer is transfer pipette that had markings every 0.1 mL