Good morning everybody
Solved
Show answers
More tips
- F Food and Cooking How to Cook a Steak with Mustard Sauce? What Kind of Meat to Choose?...
- P Photography and Videography What is lens calibration and why is it needed?...
- F Family and Home Stay Warm but Don t Overheat: What is the Optimal Temperature for Your Home During Winter?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- C Chemistry What is the answer? Please...
- C Chemistry 8th grade science by what process does carbon dioxide enter the ocean...
- C Chemistry How many nonmetal elements are on the periodic table of elements...
- C Chemistry Agroup of electrons orbiting at roughly the same distance from the nucleus...
- C Chemistry Sulfur (s) is in group 16 of the periodic table. how many valence electrons dos an atom of sulfur have?...
- C Chemistry How many grams of ca are contained in 113 ml of 0.52 m ca (oh)2 solution...
- C Chemistry Alarge value for bond energy corresponds to a more (stable/unstable) molecule, which corresponds to a (lower/higher) energy state...
- C Chemistry Find the empirical formula of a compound that contains 42g of nitrogen and 9g of hydrogen....
- C Chemistry On an x-y graph, where is the dependent variable scale (the marked increments)?...
- H History Ican t find either of these in my text book, ! which is an argument for free trade? (points : 3) it encourages international cooperation. it leads to peaceful resolutions...

Ответ:
Ответ:
the answer is 26. have a great day!
Ответ:
Answer :
(a) The concentration of is, 0.0337 M
is, 0.0337 M
(b) The concentration of is,
is, 
Solution :
(a) As per question, lead is oxidized and copper is reduced.
The oxidation-reduction half cell reaction will be,
Oxidation half reaction:
Reduction half reaction:
The balanced cell reaction will be,
Here lead (Pb) undergoes oxidation by loss of electrons, thus act as anode. Copper (Cu) undergoes reduction by gain of electrons and thus act as cathode.
First we have to calculate the standard electrode potential of the cell.
Now we have to calculate the concentration of .
.
Using Nernest equation :
where,
n = number of electrons in oxidation-reduction reaction = 2
Now put all the given values in the above equation, we get:
Therefore, the concentration of is, 0.0337 M
is, 0.0337 M
(b) As per question, lead is reduced and copper is oxidized.
The oxidation-reduction half cell reaction will be,
Oxidation half reaction:
Reduction half reaction: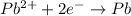
The balanced cell reaction will be,
Here Copper (Cu) undergoes oxidation by loss of electrons, thus act as anode. Lead (Pb) undergoes reduction by gain of electrons and thus act as cathode.
First we have to calculate the standard electrode potential of the cell.
Now we have to calculate the concentration of .
.
Using Nernest equation :
where,
n = number of electrons in oxidation-reduction reaction = 2
Now put all the given values in the above equation, we get:
Therefore, the concentration of is,
is, 